Pitting corrosion behavior of pure copper components in EHV/UHV DC transmission environment
-
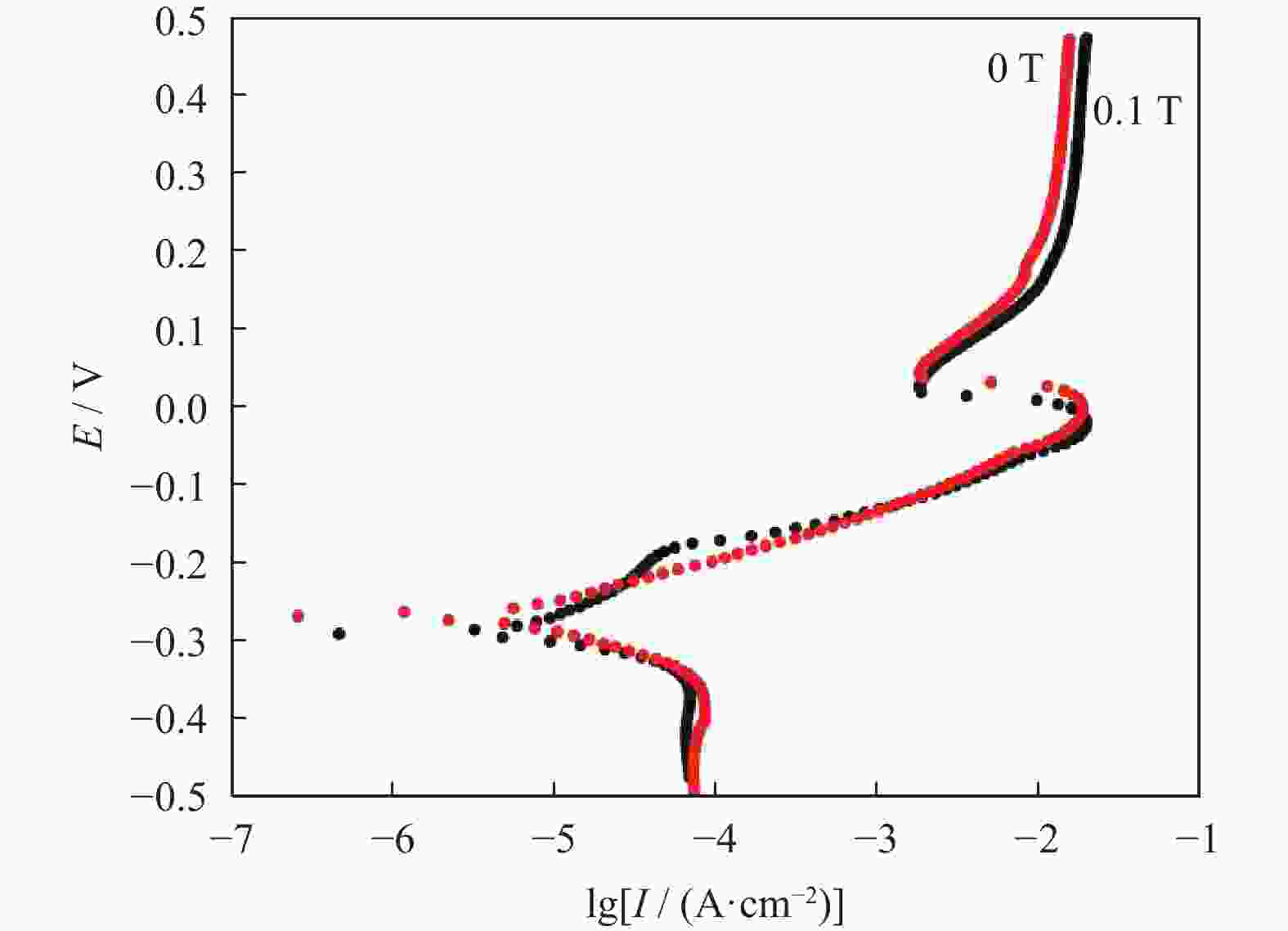
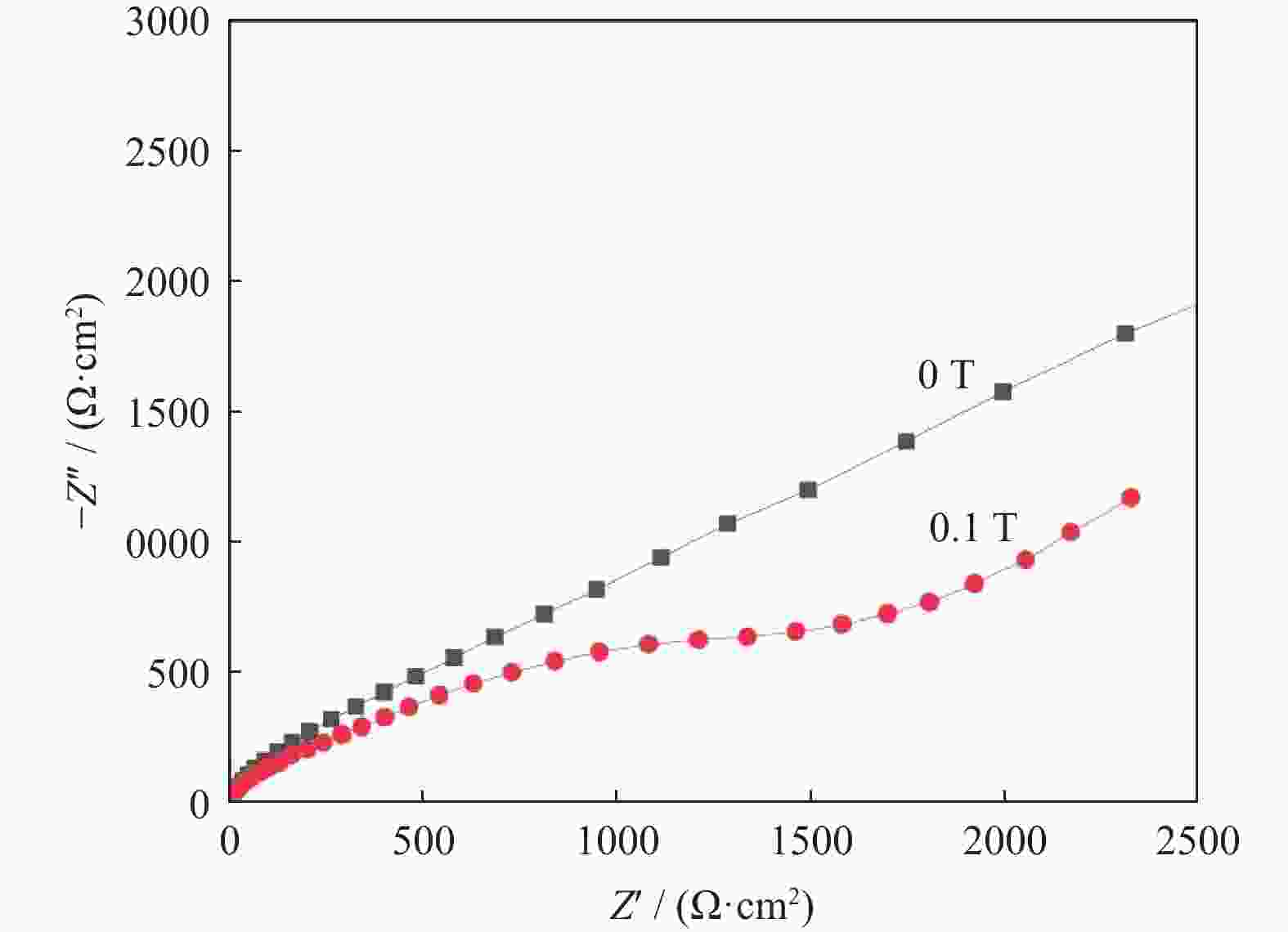
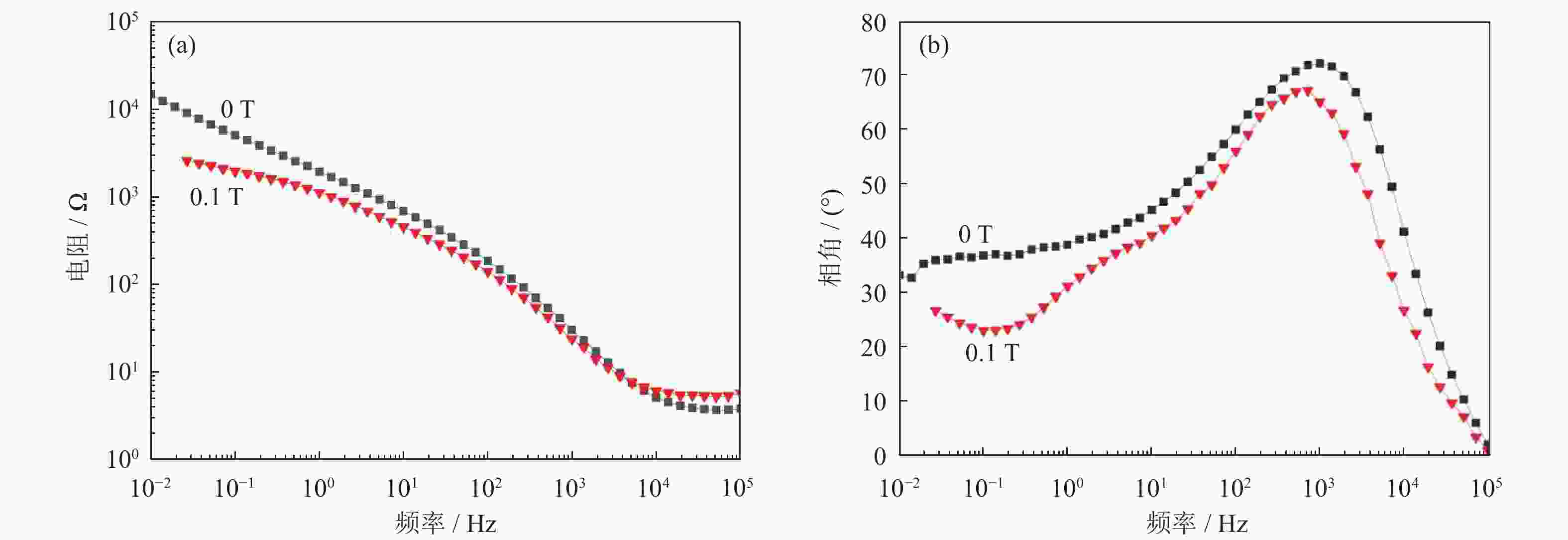
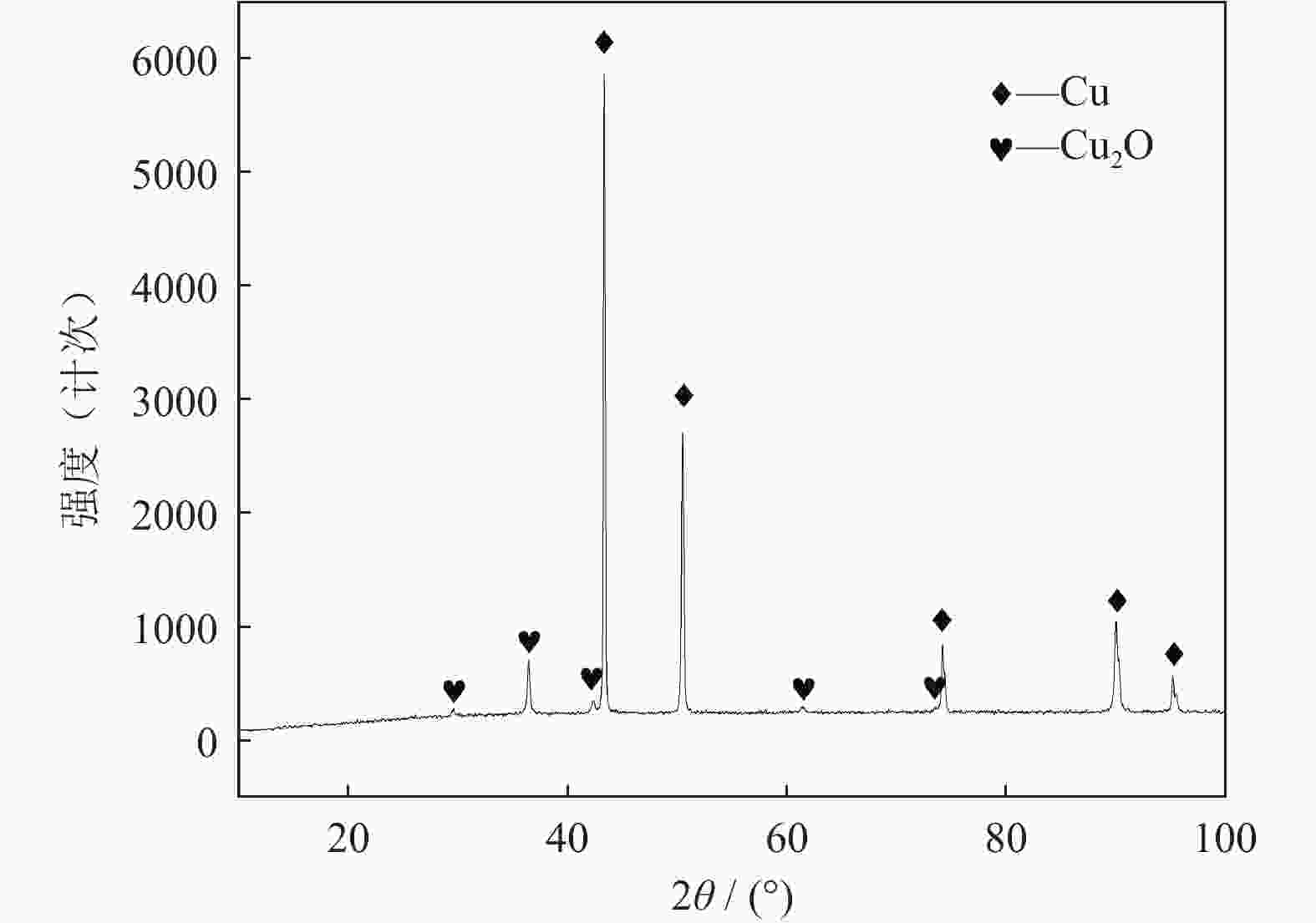
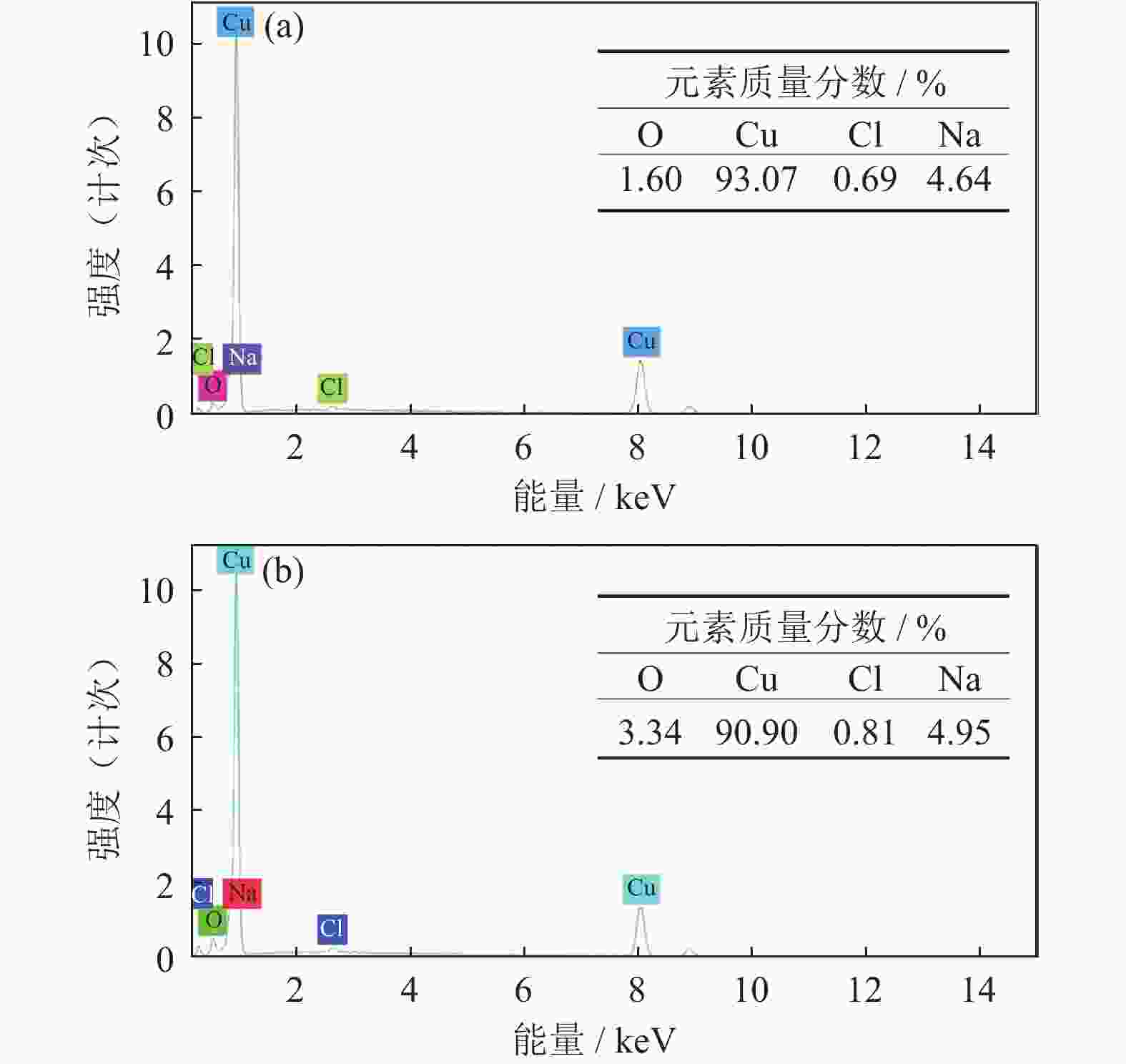
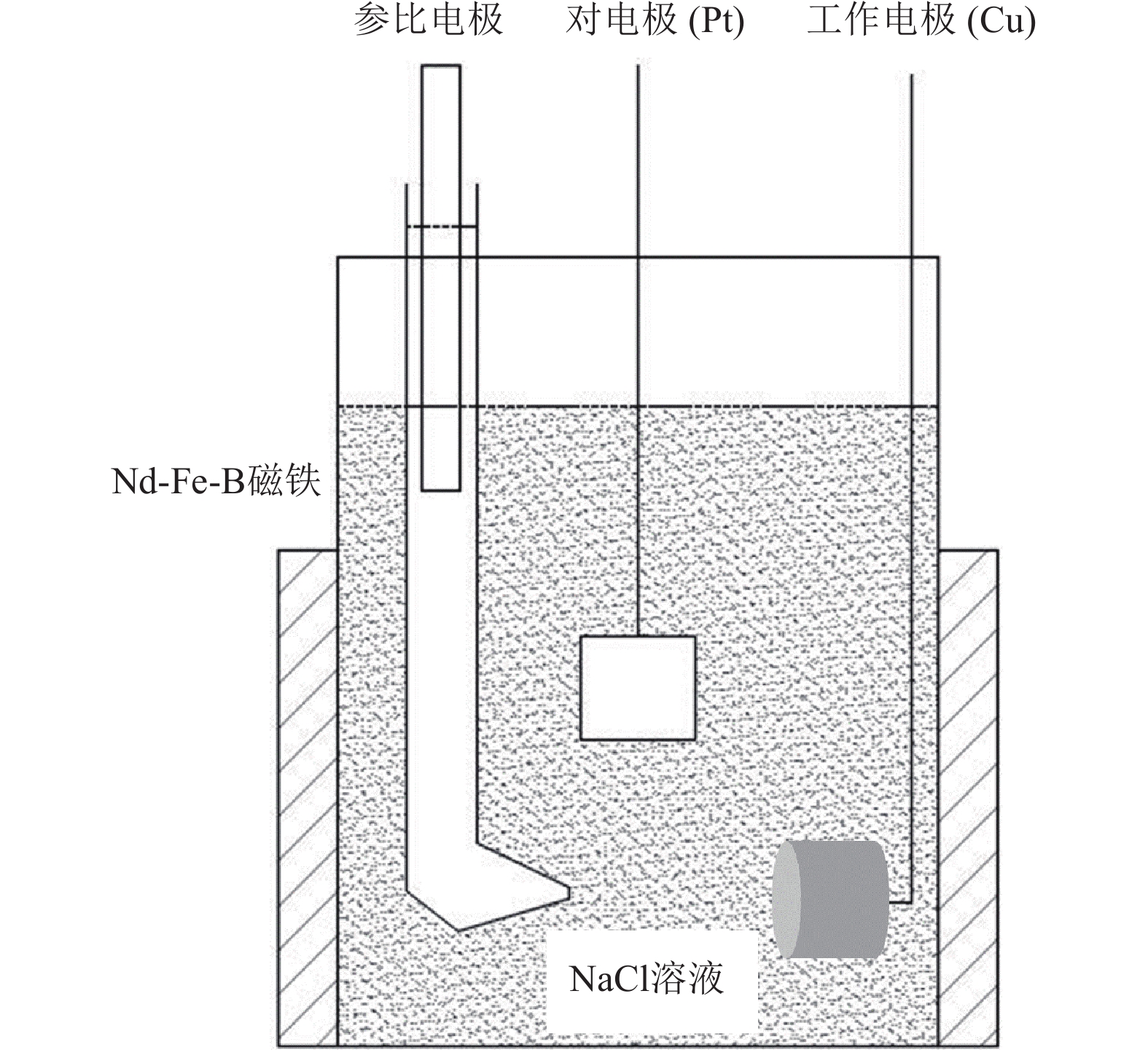
摘要: 超/特高压输电是实现全球能源互联的核心技术。作为应用最广泛的输电系统导体材料,铜及铜合金的耐蚀性被重点关注。在超/特高压直流输电过程中,周围环境存在较大磁场,导致铜部件的服役环境与普通输电环境不同。采用动电位极化、电化学阻抗谱和元素分析等方法对纯铜在超/特高压环境下的电化学腐蚀行为进行了研究。结果表明:在超/特高压环境下,纯铜在3.5%NaCl溶液(质量分数)中的极限扩散电流高于无磁场条件下的极限扩散电流,并且无磁场条件下的反应电阻比施加了0.1 T磁场条件下的反应电阻明显提高。结合电化学阻抗谱、X射线衍射及元素分析可知,0.1 T磁场会使纯铜的耐蚀性降低,主要腐蚀产物为氧化亚铜。Abstract: EHV/UHV power transmission is the core technology to realize the global energy interconnection. As the most widely used conductor materials in power transmission system, the corrosion resistance of copper and copper alloys has been paid more attention. In the process of EHV/UHV DC transmission, there is a large magnetic field in the surrounding environment, which leads to the service environment of the copper components different from that of the ordinary transmission environment. The electrochemical corrosion behavior of the pure copper under the EHV/UHV environment was studied by potential polarization, electrochemical impedance spectroscopy, and element analysis in this paper. The results show that, the limiting diffusion current of the pure copper in 3.5% NaCl solution (mass fraction) under the UHV/UHV environment is larger than that without magnetic field, and the reaction resistance without magnetic field is significantly increased compared with that under 0.1 T magnetic. Combined with the results of electrochemical impedance spectroscopy, X-ray diffraction, and elemental analysis, it is shown that the high-intensity magnetic field (0.1 T) can reduce the corrosion resistance of the pure copper, and the main corrosion product is Cu2O.
-
Key words:
- EHV/UHV DC transmission /
- magnetic field /
- pure copper /
- corrosion resistance /
- electrochemistry
-
表 1 纯铜化学成分(质量分数)
Table 1. Chemical composition of the pure copper
% Cu P Fe Zn Si Ag Ni 99.9000 0.0007 0.0018 0.0029 0.0014 0.0010 0.0022 表 2 纯铜在不同条件下的极化曲线拟合结果
Table 2. Fitting results of the polarization curves of the pure copper under the different conditions
磁感应
强度 / T腐蚀电位,
Ecorr / mV腐蚀电流,
Icorr / (μA·cm−2)极限扩散电流,
Id / (μA·cm−2)0 −267.954 0.970 13.43×103 0.1 −291.224 1.067 17.43×103 表 3 纯铜在不同条件下电化学阻抗谱拟合结果
Table 3. EIS fitting results of the pure copper under the different conditions
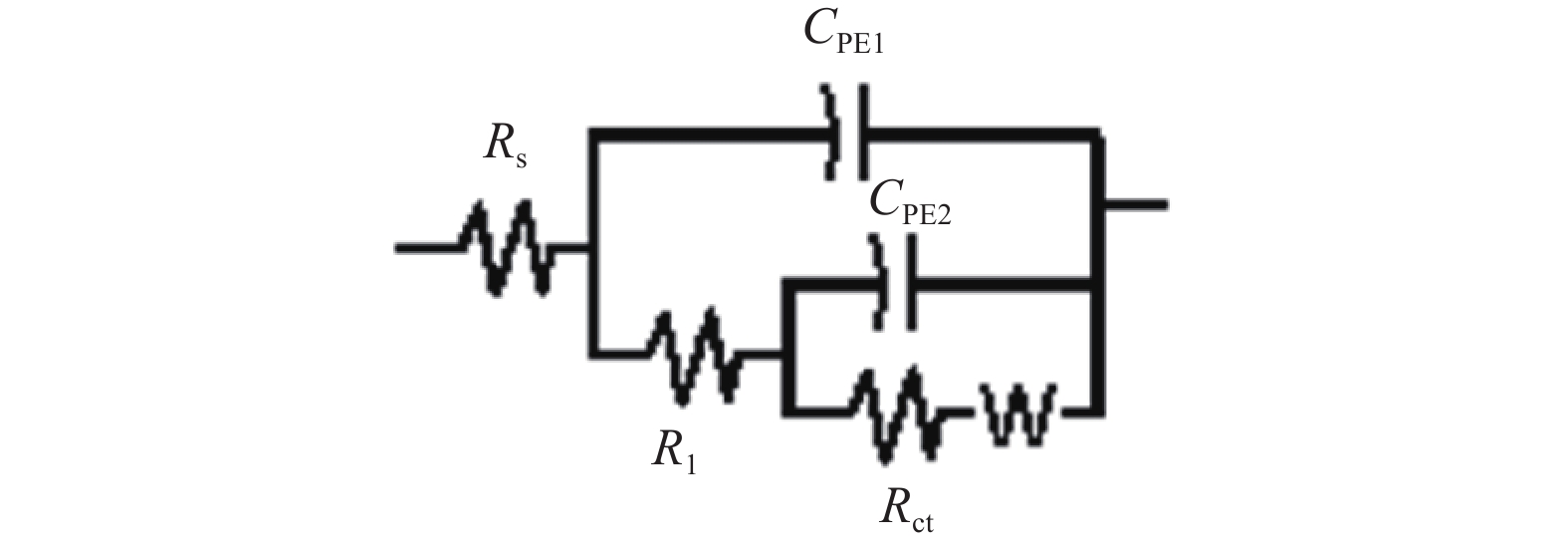
磁感应强度 / T RS / (Ω·cm2) CPE1 / F R1 / (Ω·cm2) CPE2 / F Rct / (Ω·cm2) WR / Ω 0 1.000×10−2 2.146×10−9 6.766 1.841×10−6 46.440 1.787×10−4 0.1 1.000×10−2 8.044×10−9 3.097 1.276×10−17 4.715×10−2 2.864×10−4 -
[1] Chang H, Fan J C. System design and its localization of UHVDC transmission project. Power Syst Technol, 2006, 30(16): 1 doi: 10.3321/j.issn:1000-3673.2006.16.001常浩, 樊纪超. 特高压直流输电系统成套设计及其国产化. 电网技术, 2006, 30(16): 1 doi: 10.3321/j.issn:1000-3673.2006.16.001 [2] He C W, Liu P H. Application of UHVDC transmission technology. Technol Innov Appl, 2020(20): 177何慈武, 刘鹏华. 特高压直流输电技术的应用探究. 科技创新与应用, 2020(20): 177 [3] Zhao X L, Liu Y, Wu J W, et al. Technical and economic demands of HVDC submarine cable technology for Global Energy Interconnection. Wire J Int, 2021, 54(3): 62 [4] Wang H T. Study on new protection principle of UHV DC transmission line. Electron Test, 2016(24): 51 doi: 10.3969/j.issn.1000-8519.2016.24.030王宏韬. 特高压直流输电线路保护新原理研究. 电子测试, 2016(24): 51 doi: 10.3969/j.issn.1000-8519.2016.24.030 [5] Du X Q, Yang Q S, Chen Y, et al. Galvanic corrosion behavior of copper/titanium galvanic couple in artificial seawater. Trans Nonferrous Met Soc China, 2014, 24(2): 570 doi: 10.1016/S1003-6326(14)63097-1 [6] Van Ingelgem Y, Tourwe E, Vereecken J, et al. Application of multisine impedance spectroscopy, FE-AES and FE-SEM to study the early stages of copper corrosion. Electrochim Acta, 2008, 53(25): 7523 doi: 10.1016/j.electacta.2008.01.052 [7] Wu H, Dong C C, Ding G Q, et al. Corrosion resistance of three kinds of copper alloys in first-class reverse osmosis seawater. Equip Environ Eng, 2020, 17(6): 72吴恒, 董彩常, 丁国清, 等. 铜合金在一级反渗透海水中的耐蚀性对比研究. 装备环境工程, 2020, 17(6): 72 [8] Feng Q J. Study of Electrochemical Corrosion Behaviors of Copper, Zinc and Their Alloy [Dissertation]. Wuhan: Huazhong University of Science and Technology, 2015冯秋洁. 铜、锌及其合金的电化学腐蚀研究[学位论文]. 武汉: 华中科技大学, 2015 [9] Song Q N, Tong Y, Xu N, et al. Synergistic effect between cavitation erosion and corrosion for various copper alloys in sulphide-containing 3.5% NaCl solutions. Wear, 2020, 450-451: 203258 [10] Luo J Q, Hein C, Pierson J F, et al. Localised corrosion attacks and oxide growth on copper in phosphate-buffered saline. Mater Charact, 2019, 158: 109985 doi: 10.1016/j.matchar.2019.109985 [11] Guo D, Kwok C T. Effect of pH on the corrosion behavior of tungsten-copper alloys. Corros Sci, 2020, 177: 108994 doi: 10.1016/j.corsci.2020.108994 [12] Zhang X, Wang Z H, Zhou Z H, et al. Impact of magnetic field on corrosion performance of Al–Mg alloy with different electrode potential phases. Intermetallics, 2021, 129: 107037 doi: 10.1016/j.intermet.2020.107037 [13] Wang J R, Bai Z H, Xiao K, et al. Effect of static magnetic field on mold corrosion of printed circuit boards. Bioelectrochemistry, 2020, 131: 107394 doi: 10.1016/j.bioelechem.2019.107394 [14] Fu Y Y, Xu L, Yin Y L, et al. Analysis of electromagnetic environment of ±800 kV DC transmission lines. Heilongjiang Electr Power, 2014, 36(2): 118 doi: 10.3969/j.issn.1002-1663.2014.02.006付焱燚, 徐溧, 阴酉龙, 等. ±800 kV直流输电线路电磁场环境分析. 黑龙江电力, 2014, 36(2): 118 doi: 10.3969/j.issn.1002-1663.2014.02.006 [15] Hou L W. Safety Assessment of Electromagnetic Environment for UHVDC Transmission Lines [Dissertation]. Lanzhou: Lanzhou Jiaotong University, 2017侯立伟. 特高压直流输电线路电磁环境安全评估[学位论文]. 兰州: 兰州交通大学, 2017 [16] Hulett L D, Bacarella A L, LiDonnici L, et al. Analysis of protective oxide films on copper—nickel alloys by photoelectron spectroscopy. J Electron Spectrosc Relat Phenom, 1972, 1(2): 169 [17] Chen S X, Fan D, Zhang S P. Effects of magnetic field on electrochemical corrosion behavior. Mater Prot, 2015, 48(9): 31陈散兴, 樊栋, 张三平. 磁场对电化学腐蚀行为的影响. 材料保护, 2015, 48(9): 31 [18] Hu J, Dong C F, Li X G, et al. Effects of applied magnetic field on corrosion of beryllium copper in NaCl solution. J Mater Sci Technol, 2010, 26(4): 355 doi: 10.1016/S1005-0302(10)60058-8 [19] O'Brien R N, Santhanam K S V. Electrochemical hydrodynamics in a magnetic field with laser interferometry. Electroch Acta, 1987, 32(12): 1679 doi: 10.1016/0013-4686(87)80003-8 [20] Cheng J, Pan J S, Wang T Q, et al. Micro-galvanic corrosion of Cu/Ru couple in potassium periodate (KIO4) solution. Corros Sci, 2018, 137: 184 doi: 10.1016/j.corsci.2018.03.045 [21] Zou S W, Li X G, Dong C F, et al. Electrochemical migration, whisker formation, and corrosion behavior of printed circuit board under wet H2S environment. Electrochim Acta, 2013, 114: 363 doi: 10.1016/j.electacta.2013.10.051 [22] Guo B, Zhang P, Jin Y P, et al. Effects of alternating magnetic field on the corrosion rate and corrosion products of copper. Rare Met, 2008, 27(3): 324 doi: 10.1016/S1001-0521(08)60138-2 [23] Kear G, Barker B D, Walsh F C. Electrochemical corrosion of unalloyed copper in chloride media—a critical review. Corros Sci, 2004, 46(1): 109 doi: 10.1016/S0010-938X(02)00257-3 -



 下载:
下载: