Preparation of graphene oxide composites and study on adsorption properties of copper ions
-
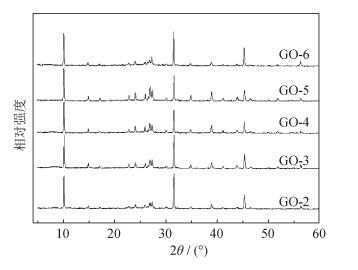
摘要: 通过超声波和磁力搅拌法制备了氧化石墨烯-4A分子筛复合材料(GO-4A), 利用X射线衍射(X-ray diffraction, XRD)、傅里叶变换红外光谱(Fourier transform-infrared spectroscopy, FT-IR) 及扫描电子显微观察(scanning electron microscopy, SEM) 等多种手段对氧化石墨烯复合材料进行表征, 并研究了氧化石墨烯与4A分子筛在GO-4A复合材料中的质量比(复合比例) 对GO-4A吸附铜离子性能的影响。结果表明: 复合比例对GO-4A物相组成影响不大; 4A分子筛的加入会降低氧化石墨烯的团聚程度, 随着复合比例增大, 复合材料的热稳定性不断提高; 当氧化石墨烯和4A分子筛的复合比例为1:5时, 复合效果最佳, 在室温条件下, 溶液pH=6时, 对铜离子的去除效率可达到98.42%。Abstract: The composites of graphene oxide-4 A molecular sieve (GO-4 A) were prepared by ultrasonic and magnetic stiring method. The graphene oxide composites were characterized by X-ray diffraction analysis (XRD), Fourier transform-infrared spectroscopy (FT-IR), and scanning electron microscopy (SEM). The effects of component ratio (i.e., the mass fractions of graphene oxide and 4 A molecular sieve in GO-4 A composites) on the adsorption of copper ions were studied. The results show that, the influence of component ratio on the phase composition of GO-4 A composites is limited. With the increase in 4 A molecular sieve content, the aggregation degree of graphene oxide is decreased, and the thermal stability of GO-4 A composites is improved. When the component ratio is 1:5, the composite effect achieves the best, the removal of copper ions (Cu2+) is up to 98.42% at the room temperature and pH = 6.
-
Key words:
- graphene oxide /
- molecular sieve /
- adsorption properties /
- copper ions
-
表 2 不同复合比例制备的GO-4A复合材料傅里叶变换红外光谱分析结果
Table 2. FT-IR analysis results of GO-4A composites prepared by different component ratios
cm-1 试样 O-H伸缩振动 O-H弯曲振动 C=C伸缩振动 C-OH伸缩振动 C-H伸缩振动 4A分子筛内部双四元环的特征振动 GO-2 3393 1643 1551 1075 934 589 GO-3 3387 1643 1555 1079 934 589 GO-4 3377 1636 1534 1065 940 590 GO-5 3382 1633 1541 1065 930 586 GO-6 3390 1640 1535 1062 — 580 -
[1] Zhang J M, Wang A N, Li H J, et al. Influence on the structure and methylene blue adsorption of graphite oxide prepared by three modified Hummers methods. Powder Metall Technol, 2018, 36(1): 16 https://www.cnki.com.cn/Article/CJFDTOTAL-FMYJ201801003.htm张建民, 王阿宁, 李红玑, 等. 三种改性Hummers法对氧化石墨结构和亚甲基蓝吸附性能的影响. 粉末冶金技术, 2018, 36(1): 16 https://www.cnki.com.cn/Article/CJFDTOTAL-FMYJ201801003.htm [2] Zhang G X. Effects of copper-coated graphite content and particle size on properties of the Cu/copper-coated graphite composite. Powder Metall Technol, 2016, 34(3): 196 doi: 10.3969/j.issn.1001-3784.2016.03.007张国玺. 石墨含量及粒度对铜-镀铜石墨复合材料性能的影响. 粉末冶金技术, 2016, 34(3): 196 doi: 10.3969/j.issn.1001-3784.2016.03.007 [3] Chabot V, Higgins D, Yu A, et al. A review of graphene and graphene oxide sponge: material synthesis and applications to energy and the environment. Energy Environ Sci, 2014, 7(5): 1564. doi: 10.1039/c3ee43385d [4] Liu L. Study on the Preparation of Montmorillonite-Pillared Graphene Oxide Composites and the Adsorption of Pollutants in Water[Dissertation]. Guangzhou: Jinan University, 2015刘露. 蒙脱土柱撑氧化石墨烯的制备及对水中污染物的吸附性能, 广州: 暨南大学, 2015 [5] Wang H, Yuan X, Wu Y, et al. Adsorption characteristics and behaviors of graphite oxide for Zn(Ⅱ)removal from aqueous solution. Appl Surf Sci, 2013, 279(8): 432. http://smartsearch.nstl.gov.cn/paper_detail.html?id=a682120f4c43aee4b45698e359dbb952 [6] Perreault F, Fonseca de F A, Elimelech M. Environmental applications of graphene-based nanomaterials. Chem Soc Rev, 2015, 44(16): 5861. doi: 10.1039/C5CS00021A [7] Xiao L, Wang W L, Yu S L, et al. Graphene-containing composite materials for water treatment. Prog Chem, 2013, 25(2-3): 419 https://www.cnki.com.cn/Article/CJFDTOTAL-HXJZ2013Z1024.htm肖蓝, 王炜龙, 于水利, 等. 石墨烯及其复合材料在水处理中的应用. 化学进展, 2013, 25(2-3): 419 https://www.cnki.com.cn/Article/CJFDTOTAL-HXJZ2013Z1024.htm [8] Liu F, Chung S, Oh G, et al. Three-dimensional graphene oxide nanostructure for fast and efficient water-soluble dye removal. ACS Appl Mater Interfaces, 2012, 4(2): 922. doi: 10.1021/am201590z [9] Reddy D A, Ma R, Choi M Y, et al. Reduced graphene oxide wrapped ZnS-Ag2S ternary composites synthesized via hydrothermal method: Applications in photocatalyst degradation of organic pollutants. Appl Surf Sci, 2015, 324: 725. doi: 10.1016/j.apsusc.2014.11.026 [10] Zhao G, Ren X, Gao X, et al. Removal of Pb(Ⅱ)ions from aqueous solutions on few-layeredgraphene oxide nanosheets. J Dalton Transactions, 2011, 40(41): 10945. doi: 10.1039/c1dt11005e [11] Gui H, Tan W, Li B, et al. Removal ammonia-nitrogen from medium-low concentration wastewater by 4A zeolite molecular sieve. Chin J Environ Eng, 2014, 8(5): 1944 https://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201405042.htm桂花, 谭伟, 李彬, 等. 4A沸石分子筛处理中低浓度氨氮废水. 环境工程学报, 2014, 8(5): 1944 https://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201405042.htm [12] Dang R. Experimental Study on Removing Phosphorus by Natural and Modified Zeolite[Dissertation]. Lanzhou: Lanzhou Jiaotong University, 2015党瑞. 天然及改性沸石去除水中磷的实验研究, 兰州: 兰州交通大学, 2015 [13] Li Y. Ion Exchange and Adsorption Performance of Zeolite[Dissertation]. Taiyuan: Taiyuan University of Technology, 2009李妍. 分子筛分子筛离子交换及其吸附性能研究, 太原: 太原理工大学, 2009 [14] Ao X, Liu H, Wang Q, et al. Adsorption of Cd(Ⅱ)in aqueous solution by granular 4A molecular sieve/attapulgite. Technol Water Treat, 2017(9): 62 https://www.cnki.com.cn/Article/CJFDTOTAL-SCLJ201709013.htm敖翔, 刘红, 汪茜, 等. 4A分子-凹凸棒土颗粒对水中Cd(Ⅱ)的吸附. 水处理技术, 2017(9): 62 https://www.cnki.com.cn/Article/CJFDTOTAL-SCLJ201709013.htm [15] Su D F. Synthesis and Adsorption Properties of Metal-Organic Frameworks(MIL-53)/4A Zeolite Material[Dissertation]. Beijing: Beijing Jiaotong University, 2017苏东方. 金属有机骨架(MIL-53)/4A沸石复合材料的制备及其吸附性能的研究, 北京: 北京交通大学, 2017 [16] Peng L Q, Xie J H, Guo C, et al. Review of characterization methods of graphene. J Funct Mater, 2013, 44(21): 3055 doi: 10.3969/j.issn.1001-9731.2013.21.001彭黎琼, 谢金花, 郭超, 等. 石墨烯的表征方法. 功能材料, 2013, 44(21): 3055 doi: 10.3969/j.issn.1001-9731.2013.21.001 -




 下载:
下载: