-
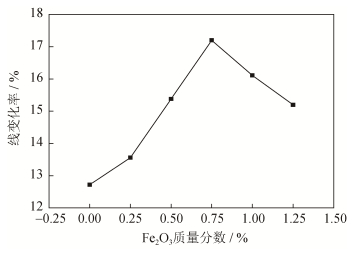
摘要: 以分析纯Ca(OH)2和m-ZrO2为原料, 按物质的量1:1进行配料, 添加不同质量分数Fe2O3粉末作为添加剂, 经充分混合后压制成ø20 mm×20 mm圆柱试样, 再经1600℃保温3 h煅烧制备得到锆酸钙陶瓷试样(CaZrO3)。利用显气孔体密测定仪、X射线衍射仪及扫描电子显微镜分析Fe2O3粉末添加剂对CaZrO3陶瓷材料烧结性能、物相组成及微观结构的影响。结果表明:当没有添加Fe2O3粉末时, 试样烧结前后线变化率为12.72%, 体积密度为3.92g·cm-3, 显气孔率为14.9%, CaZrO3晶粒尺寸为4.22μm; 当加入质量分数0.75%Fe2O3粉末时, 试样烧结前后线变化率为17.20%, 体积密度为4.68 g·cm-3, 显气孔率为6.8%, CaZrO3晶粒尺寸为5.21μm。Abstract: Using analytical Ca(OH)2 and m-ZrO2 as the raw materials in the mole ratio of 1:1, the compacted cylinder samples of calcium zirconate (CaZrO3) ceramics in the dimension of ø20 mm × 20 mm were synthetized by sintering at 1600 ℃ for 3 h, adding the Fe2O3 powders in different mass fractions as the additive agent.The effects of Fe2O3 additive agent on sintering properties, phase composition, and microstructures of CaZrO3 ceramics were studied by apparent porosity density determinator, X-ray diffraction (XRD) diffractometer, and scanning electron microscope (SEM).The results show that, without the addition of Fe2O3, the sharking of CaZrO3 samples is 12.72%, the bulk density and apparent porosity of sintered CaZrO3 samples at 1600 ℃ are 3.92 g·cm-3 and 14.9%, respectively, and the grain size of CaZrO3 is only 4.22 μm.With the addition of Fe2O3 in mass fraction of 0.75%, the sharking of CaZrO3 samples is 17.20%, the bulk density and apparent porosity of sintered CaZrO3 samples at 1600 ℃ are 4.68 g·cm-3 and 6.8%, respectively, and the grain size of CaZrO3 reaches to 5.21 μm.
-
Key words:
- calcium zirconate ceramics /
- additive agent /
- bulk density /
- apparent porosity /
- phase composition /
- microstructures
-
表 1 Fe2O3质量分数与CaZrO3晶粒直径的关系
Table 1. Relationship between CaZrO3 particle diameter and the addition content of Fe2O3 by mass
Fe2O3质量分数/ % 0 0.25 0.50 0.75 1.00 1.25 CaZrO3晶粒直径/ μm 4.22 4.45 4.98 5.21 5.04 4.89 表 2 图 6晶界处(位置1)和晶粒处(位置2)能谱分析
Table 2. Energy spectrum analysis of crystal boundary (position 1) and crystal particle (position 2) in Fig.6
元素 晶界处成分(位置1) 晶粒处成分(位置2) 质量分数/ % 原子数分数/ % 质量分数/ % 原子数分数/ % O 32.13 66.66 38.25 72.53 Ca 18.29 15.14 16.92 12.78 Zr 48.90 17.79 44.82 14.87 Fe 0.68 0.40 0 0 总计 100 100 100 100 -
[1] Yajima T, Kazeoka H, Yogo T, et al. Proton conduction in sintered oxides based on CaZrO3. Solid State Ionics, 1991, 47(3): 271. http://www.sciencedirect.com/science/article/pii/016727389190249B [2] van Rij L, Winnubst L, Jun L, et al. Analysis of the preparation of in-doped CaZrO3 using a peroxo-oxalate complexation method. J Mater Chem, 2000, 10(11): 2515. doi: 10.1039/b003840g [3] Liao Y, Zhu W, Li X M, et al. Preparation of Li(2‒x)(MoO4)2:Eux phosphors by sol‒gel process and their characterization. Mater Rev, 2007, 21(Special issue Ⅷ): 321 https://www.cnki.com.cn/Article/CJFDTOTAL-CLDB2007S1098.htm廖勇, 朱伟, 黎学明, 等.溶胶-凝胶法制备Li(MoO4)2: Eu及表征.材料导报, 2007, 21(专辑Ⅷ): 321 https://www.cnki.com.cn/Article/CJFDTOTAL-CLDB2007S1098.htm [4] Ling J D. Introduction of CaZO3 refractories. Bull Chin Ceram Soc, 1986, 5(3): 26 https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT198603004.htm凌继栋.锆酸钙耐火材料简介.硅酸盐通报, 1986, 5(3): 26 https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT198603004.htm [5] Hang J D, Wen Z Y, Zhang J C, et al. CaZrO3 based high temperature proton conductors. Prog Chem, 2012, 24(9): 1845 https://www.cnki.com.cn/Article/CJFDTOTAL-HXJZ201209023.htm韩金铎, 温兆银, 张敬超, 等.锆酸钙基高温质子导体材料.化学进展, 2012, 24(9): 1845 https://www.cnki.com.cn/Article/CJFDTOTAL-HXJZ201209023.htm [6] Liang L P, Gao Y B, Chen S Y. Preparation of CaO-ZrO2ultrafine powder by coprecipitation-supercritical fluid drying method and investigation on its sintering behaviour. Bull Chin Ceram Soc, 1998, 17(3): 17 https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT803.003.htm梁丽萍, 高荫本, 陈诵英.共沉淀-超临界流体干燥法合成CaO-ZrO2复合氧化物超微粉体及其烧结性能研究.硅酸盐通报, 1998, 17(3): 17 https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT803.003.htm [7] Li W, Zhou G J, Zhang A Y, et al. Preparation and luminescence properties of rare earth-doped calcium zirconate nanocrystals. J Chin Ceram Soc, 2011, 39(11): 1729 https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB201111005.htm李玮, 周广军, 张爱玉, 等.稀土离子掺杂锆酸钙纳米晶的制备及发光性质.硅酸盐学报, 2011, 39(11): 1729 https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB201111005.htm [8] Zhang W N, Deng N, Liang W J, et al. Effect of CuO on the sintering properties of dolomite. Powder Metall Technol, 2016, 34(4): 277 doi: 10.3969/j.issn.1001-3784.2016.04.008张汪年, 邓宁, 梁伟杰, 等. CuO对白云石烧结性能的影响.粉末冶金技术, 2016, 34(4): 277 doi: 10.3969/j.issn.1001-3784.2016.04.008 [9] You J G, Zhang G D, Jin Y L, et al. Effect of SiO2 on structure and properties of CaZO3 material. J Synth Cryst, 2014, 43(5): 1280 doi: 10.3969/j.issn.1000-985X.2014.05.044游杰刚, 张国栋, 金永龙, 等.二氧化硅对锆酸钙材料结构及性能的影响.人工晶体学报, 2014, 43(5): 1280 doi: 10.3969/j.issn.1000-985X.2014.05.044 [10] You J G, Zhang G D, Gao P L, et al. Effect of Al2O3 addition on composition and structure of CaZrO3synthesized from ZrO2 and CaCO3. Trans Mater Heat Treat, 2014, 35(5): 39 https://www.cnki.com.cn/Article/CJFDTOTAL-JSCL201405008.htm游杰刚, 张国栋, 高配亮, 等.氧化铝对锆酸钙材料组成和结构的影响.材料热处理学报, 2014, 35(5): 39 https://www.cnki.com.cn/Article/CJFDTOTAL-JSCL201405008.htm [11] Gabal M A, Al-Iuhaibi R S, Al Angari Y M. Mn-Zn nano-crystalline ferrites synthesized from spent Zn-Cbatteries using novel gelatin method. J Hazard Mater, 2013, 246-247: 227. doi: 10.1016/j.jhazmat.2012.12.026 [12] Zhang W N, Deng N, Wang L. Effect of CuO addition on the properties of CaZrO3. Powder Metall Technol, 2018, 36(1): 26 doi: 10.19591/j.cnki.cn11-1974/tf.2018.01.005张汪年, 邓宁, 王利. CuO对锆酸钙烧结性能的影响.粉末冶金技术, 2018, 36(1): 26 doi: 10.19591/j.cnki.cn11-1974/tf.2018.01.005 [13] Yuan C, Zhao H Z, Zhang H, et al. Effect of ferric oxide addition on sintering property and hydration resistance of dolomite clinker. Refractories, 2011, 45(4): 265 doi: 10.3969/j.issn.1001-1935.2011.04.006袁聪, 赵惠忠, 张寒, 等.铁红对白云石熟料烧结性和抗水化性的影响.耐火材料, 2011, 45(4): 265 doi: 10.3969/j.issn.1001-1935.2011.04.006 -




 下载:
下载: