-
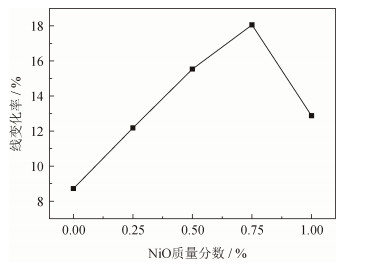
摘要: 以天然菱镁矿为原料,添加不同质量分数的NiO粉末,采用高温煅烧法制备了镁砂。利用显气孔体密测定仪、X射线衍射仪、扫描电子显微镜分析NiO粉末掺杂对镁砂熟料烧结性能、物相组成、微观结构的影响。结果表明,在菱镁矿中添加NiO粉末有利于提高镁砂烧结致密性,促进MgO晶粒长大。当不添加NiO粉末时,试样在1600℃烧结前后线变化率为8.72%,体积密度为2.96 g·cm-3,显气孔率为19.2%,MgO晶粒尺寸为1.36 μm;当加入质量百分数为0.75% NiO粉末时,试样烧结前后线变化率为18.06%,制备镁砂体积密度为3.29 g·cm-3,显气孔率为11.5%,MgO晶粒尺寸为3.81 μm。在1600℃对添加NiO粉末的菱镁矿进行烧结,NiO可以与MgO完全固溶,引起MgO晶格畸变,降低晶体活化能,提高MgO烧结致密性能。但添加过多NiO,在短时间内无法与MgO完全固溶,妨碍烧结相的直接接触,影响传质过程,导致样品烧结性能降低。Abstract: The magnesite doped by NiO powders in different mass fractions was prepared by high-temperature calcination using natural magnesite as the raw materials in this study. The effects of NiO doping on the sintered properties, phase composition, and microstructures of magnesite were characterized by apparent porosity tester, X-ray diffractometer, and scanning electron microscope. The results show that, the NiO doping can improve the compactness of magnesite and promote the growth of MgO grain. Without the NiO doping, the line rate, bulk density, and apparent porosity of the magnesite after sintering at 1600℃ are 8.72%, 2.96 g·cm-3, and 19.2%, respectively, and the particle size of MgO is 1.36 μm. The line rate, bulk density, and apparent porosity of the magnesite doped by NiO powders in the mass fraction of 0.75% after sintering at 1600℃ are 18.06%, 3.29 g·cm-3, and 11.5%, respectively, and the particle size of MgO reaches to 3.81 μm. The sintered magnesite doped by NiO powders at 1600℃ can lead to the complete solid solution bewteen MgO and NiO, cause the lattice distortion of MgO, reduce the activation energy of grain growth, and consequentially promote the sintering compactness of the magnesite. But, the over-dose NiO doping may result in the partial solid solution bewteen MgO and NiO within a short time, impede the direct contact of sintered phases, obstruct the mass transfer process, and consequentially lower the sintering compactness of the magnesite.
-
Key words:
- magnesite /
- doping /
- sintering properties /
- phase composition /
- microstructures
-
表 1 天然菱镁矿石的化学成分(质量分数)
Table 1. Chemical component of the natural magnesite %
MgO CaO Fe2O3 SiO2 Al2O3 灼烧量 45.23 1.69 0.84 1.03 0.13 51.08 表 2 NiO质量分数与镁砂MgO晶粒尺寸的关系
Table 2. Relationship between the content of NiO by mass and MgO grain size of magnesite clinker
NiO质量分数/% MgO晶粒尺寸/μm 0 1.36 0.25 2.58 0.50 3.19 0.75 3.81 1.00 3.55 表 3 镁砂中MgO的晶格常数
Table 3. Lattice constant of MgO of magnesia clinker
NiO质量分数/% 0 0.25 0.50 0.75 1.00 MgO晶格常数/nm 0.420202 0.420286 0.420408 0.420558 0.420560 -
[1] Yuan Y. High-purity dead-burned magnesia made from magnesite powder. China Non-met Min Ind Her, 2003, 36(5): 24 doi: 10.3969/j.issn.1007-9386.2003.05.007袁锐. 用菱镁矿粉矿制备高纯镁砂的研究. 中国非金属矿工业导刊, 2003, 36(5): 24 doi: 10.3969/j.issn.1007-9386.2003.05.007 [2] Li Z, Zhang S, Lee W E. Improving the hydration resistance of lime-based refractory materials. Int Mater Rev, 2008, 53(1): 1 doi: 10.1179/174328007X212508 [3] Chen M, Lu C Y, Yu J K. Improvement in performance of MgO-CaO refractories by addition of nano-sized ZrO2. J Eur Ceram Soc, 2007, 27(16): 4633 http://www.sciencedirect.com/science/article/pii/S0955221907003317 [4] Yeprem H A. Effect of iron oxide addition on the hydration resistance and bulk density of doloma. J Eur Ceram Soc, 2007, 27(2-3): 1651 doi: 10.1016/j.jeurceramsoc.2006.05.010 [5] Li H, Su L, Yu J K. Investigation on process flow of high-density magnesia. J Northeastern Univ Nat Sic, 2007, 28(3): 381 doi: 10.3321/j.issn:1005-3026.2007.03.020李环, 苏莉, 于景坤. 高密度烧结镁砂的研究. 东北大学学报(自然科学版), 2007, 28(3): 381 doi: 10.3321/j.issn:1005-3026.2007.03.020 [6] Zhan D, Huang L, Xiao Z A, et al. Preparation of nanometer magnesium oxide powder by rheology phase-precursor method. Chem Reag, 2007, 29(3): 141 doi: 10.3969/j.issn.0258-3283.2007.03.005占丹, 黄琳, 肖作安, 等. 流变相-前驱物法制备纳米氧化镁粉体. 化学试剂, 2007, 29(3): 141 doi: 10.3969/j.issn.0258-3283.2007.03.005 [7] Zhu Y X, Zhen R J, Liu X J, et al. Preparation and characterization of MgO nanopowder. J Xiamen Univ Nat Sic, 2001, 40(6): 1256 doi: 10.3321/j.issn:0438-0479.2001.06.015朱亚先, 曾人杰, 刘新锦, 等. MgO纳米粉制备及表征. 厦门大学学报(自然科学版), 2001, 40(6): 1256 doi: 10.3321/j.issn:0438-0479.2001.06.015 [8] Gui M X. Effect of NiO on slaking resistance of magnesia clinker. Foreign Refract, 2005, 30(3): 35 https://www.cnki.com.cn/Article/CJFDTOTAL-GWLH200503010.htm桂明玺. 添加氧化镍对提高镁砂的抗水化性能所起的效果. 国外耐火材料, 2005, 30(3): 35 https://www.cnki.com.cn/Article/CJFDTOTAL-GWLH200503010.htm [9] Zheng Z Q, Li H Y. Rare Earth Functional Materials. Beijing: Chemical Industry Press, 2003郑子樵, 李红英. 稀土功能材料. 北京: 化学工业出版社, 2003 [10] Liu L, Wang Z F, He J P, et al. Effects of Y2O3 and CeO2 on sintering and microstructure of magnesite. Refractories, 2012, 46(5): 340 https://www.cnki.com.cn/Article/CJFDTOTAL-LOCL201205007.htm刘磊, 王周福, 何俊鹏, 等. Y2O3和CeO2对镁砂烧结性能及显微结构的影响. 耐火材料, 2012, 46(5): 340 https://www.cnki.com.cn/Article/CJFDTOTAL-LOCL201205007.htm [11] Wang Z F, Xu Z W, Zhang B G, et al. Effect of mixture of rare earth oxides on microstructure and properties of magnesia refractory. Rare Met Mater Eng, 2007, 36(Suppl 2): 373 https://www.cnki.com.cn/Article/CJFDTOTAL-COSE2007S2109.htm王周福, 徐自伟, 张保国, 等. 混合稀土氧化物对镁质耐火材料结构与性能的影响. 稀有金属材料与工程, 2007, 36(增刊2): 373 https://www.cnki.com.cn/Article/CJFDTOTAL-COSE2007S2109.htm [12] Chaudhuri M, Banerjee G, Kumar A, e al. Secondary phases in natural magnesite sintered with addition of titania, ilmenite and zirconia. J Mater Sci, 1999, 34(23): 5821 doi: 10.1023/A:1004718503991 [13] Lee Y B, Park H C, Oh K D, et al. Sintering and microstructure development in the system MgO–TiO2. J Mater Sci, 1998, 33(17): 4321 doi: 10.1023/A%3A1004443728590 [14] Martinac V, Labor M, Petric N. Effect of TiO2, SiO2 and Al2O3 on properties of sintered magnesium oxide from sea water. Mater Chem Phys, 1996, 46(1): 23 http://www.sciencedirect.com/science/article/pii/0254058496801258 [15] Zhang W N, Wang L, Deng N, et al. Effect of CoO addition on the preparation of magnesia by magnesite. Powder Metall Technol, 2018, 36(4): 292 doi: 10.19591/j.cnki.cn11-1974/tf.2018.04.009张汪年, 王利, 邓宁, 等. CoO对菱镁矿制备镁砂性能的影响. 粉末冶金技术, 2018, 36(4): 292 doi: 10.19591/j.cnki.cn11-1974/tf.2018.04.009 -




 下载:
下载: