-
摘要: 纳米金刚石具有金刚石和纳米材料的双重特性,由于心部–表面的sp3/sp2杂化结构以及丰富的表面悬键和官能团,使其在研磨抛光、减磨润滑、复合材料强化、药物载体、荧光吸收等领域有着广阔的应用前景。纳米金刚石多以数百纳米尺度的颗粒团聚,需要采用各种手段和方法分散至不同的液相体系中加以应用。本文对国内外纳米金刚石分散方法进行了总结,研究和分析了机械法、无机化学法、高能量场处理法以及表面有机化学改性法等分散手段的原理及特点。Abstract: Nanodiamond has the dual characteristics of the diamond and the nano-materials. Due to the sp3/sp2 hybrid structure of core and surface and the abundant surface dangling bonds and functional groups, the nanodiamond shows the broad application in the fields of lapping and polishing, antifriction and lubrication, composite material reinforcement, drug delivery, and fluorescence absorption. The nanodiamond particles agglomerate in hundreds of nanometers and need to be dispersed into the different liquid phase systems by various means. The research on the dispersion methods of the nanodiamond was summarized at home and abroad in this paper, and the principle and characteristics of the dispersion methods as the mechanical method, the inorganic chemical method, the high energy field treatment, and the surface organic chemical modification were analysed.
-
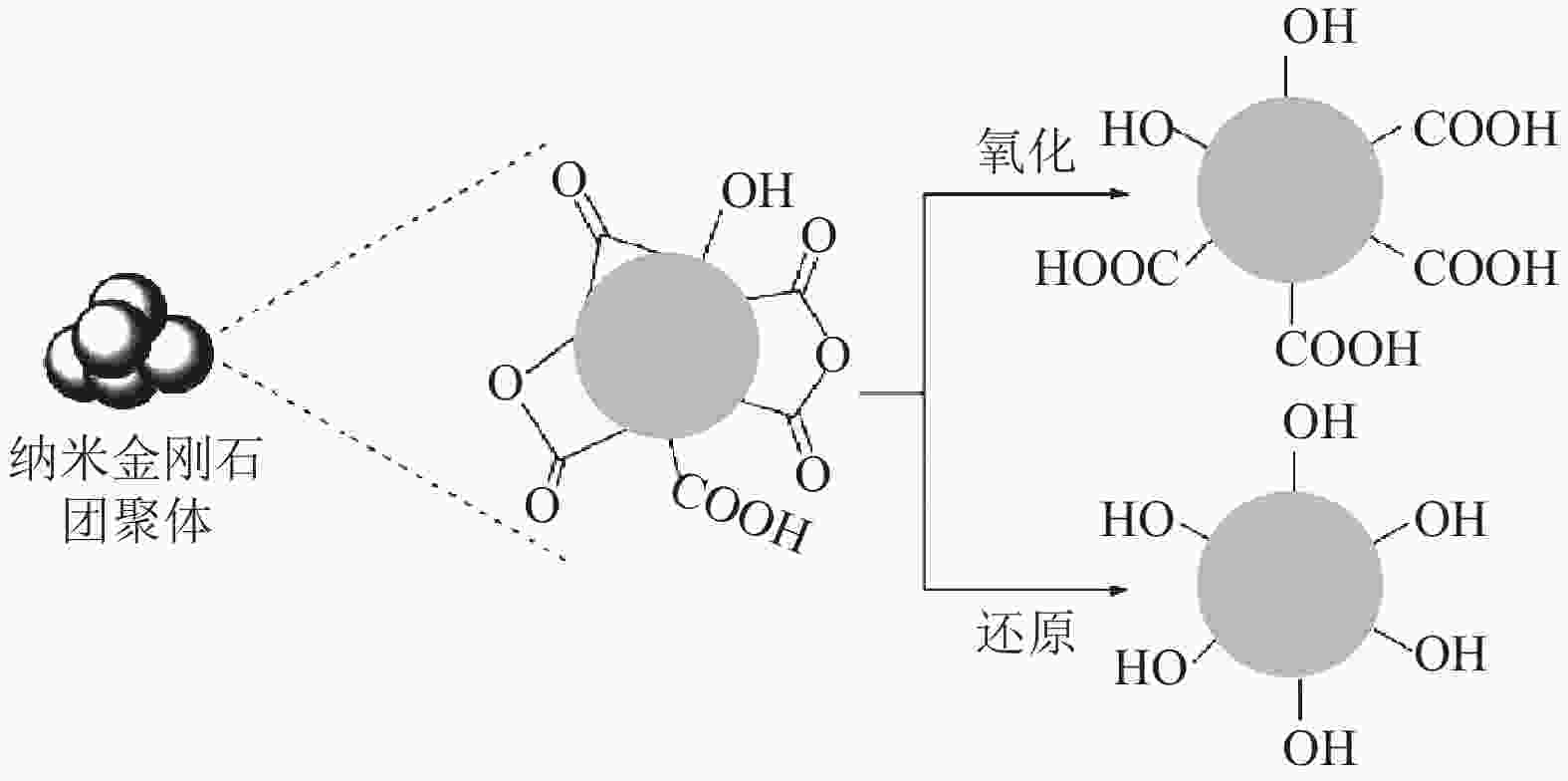
图 1 单个纳米金刚石颗粒结构示意图:(a)氧化纯化后单个粒径约5 nm的纳米金刚石结构;(b)sp2碳形成链和石墨斑;(c)纳米金刚石表面原子含氧官能团;(d)由高度有序金刚石核心组成的纳米金刚石[8]
Figure 1. Structure schematic diagram of the single nanodiamond particle: (a) the single nanodiamond structure with the paticle size of 5 nm after oxidative purification; (b) the sp2 carbon chains and the graphitic patches; (c) the oxygen-containing groups on the atom surfaces of nanodiamond; (d) the nanodiamond made up of the highly ordered diamond core[8]
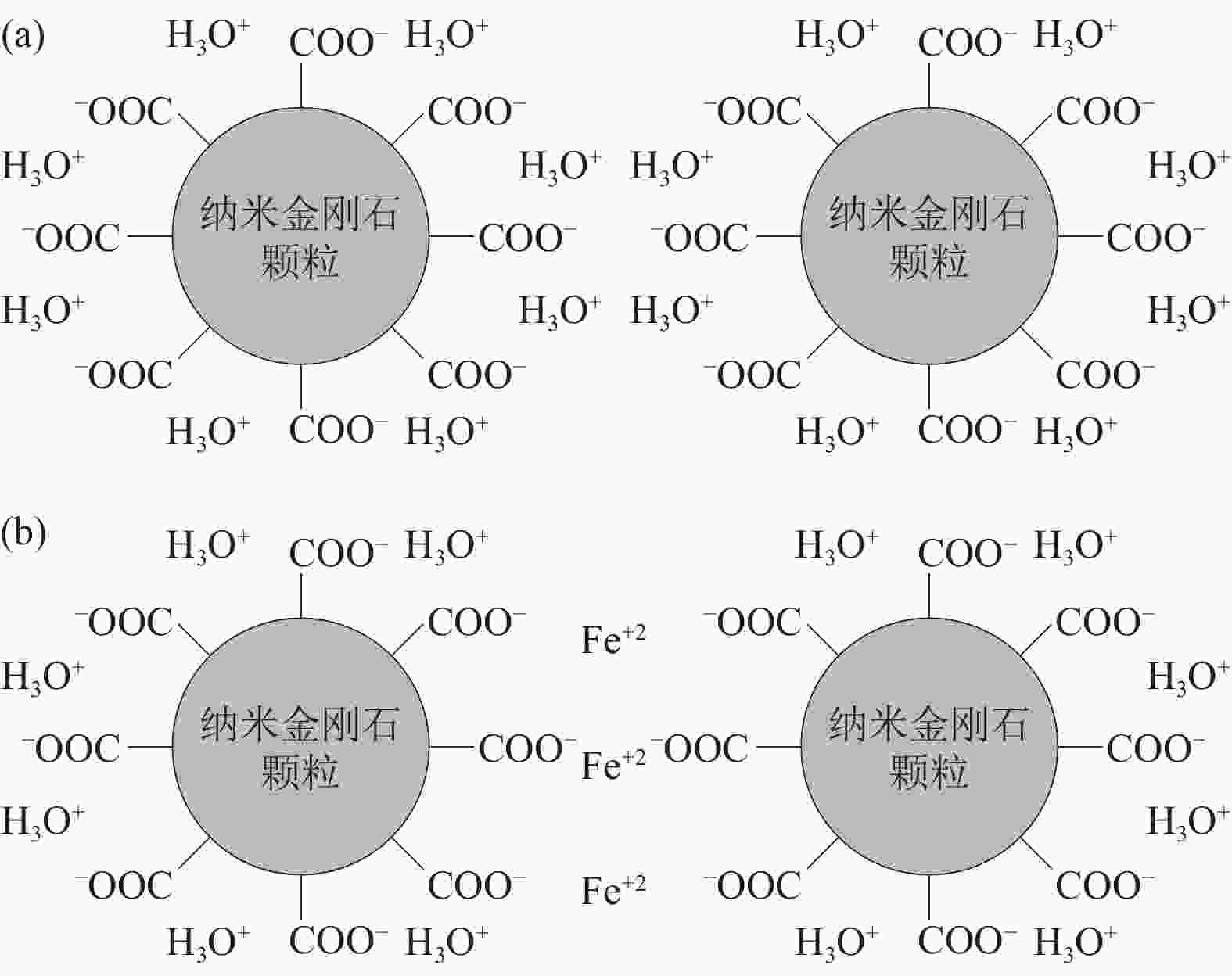
图 3 酸洗纳米金刚石示意图:(a)纳米金刚石颗粒表面的羧基–COOH双电层;(b)纳米金刚石颗粒在Fe2+作用下连接单个颗粒形成的桥接引起团聚[21]
Figure 3. Schematic diagram of the acid pickling for the dispersion of nanodiamonds: (a) the double electric layer formation of carboxyl–COOH at the nanodiamond particle surface; (b) the agglomeration by bridge bonding in the presence of iron ions of the nanodiamond particle[21]
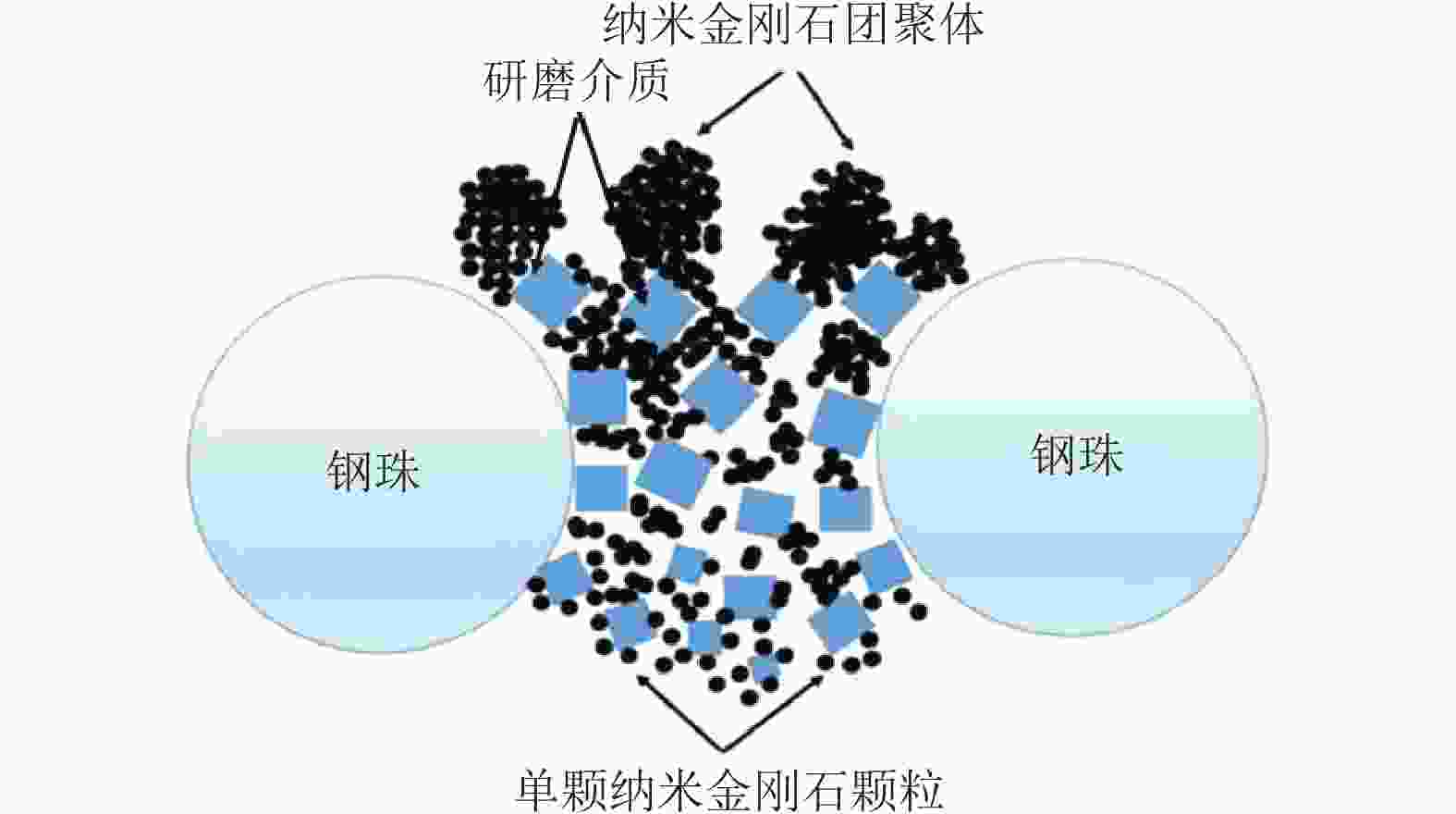
表 1 高能球磨分散纳米金刚石技术
Table 1. Dispersion method of the nanodiamond by high energy ball mill
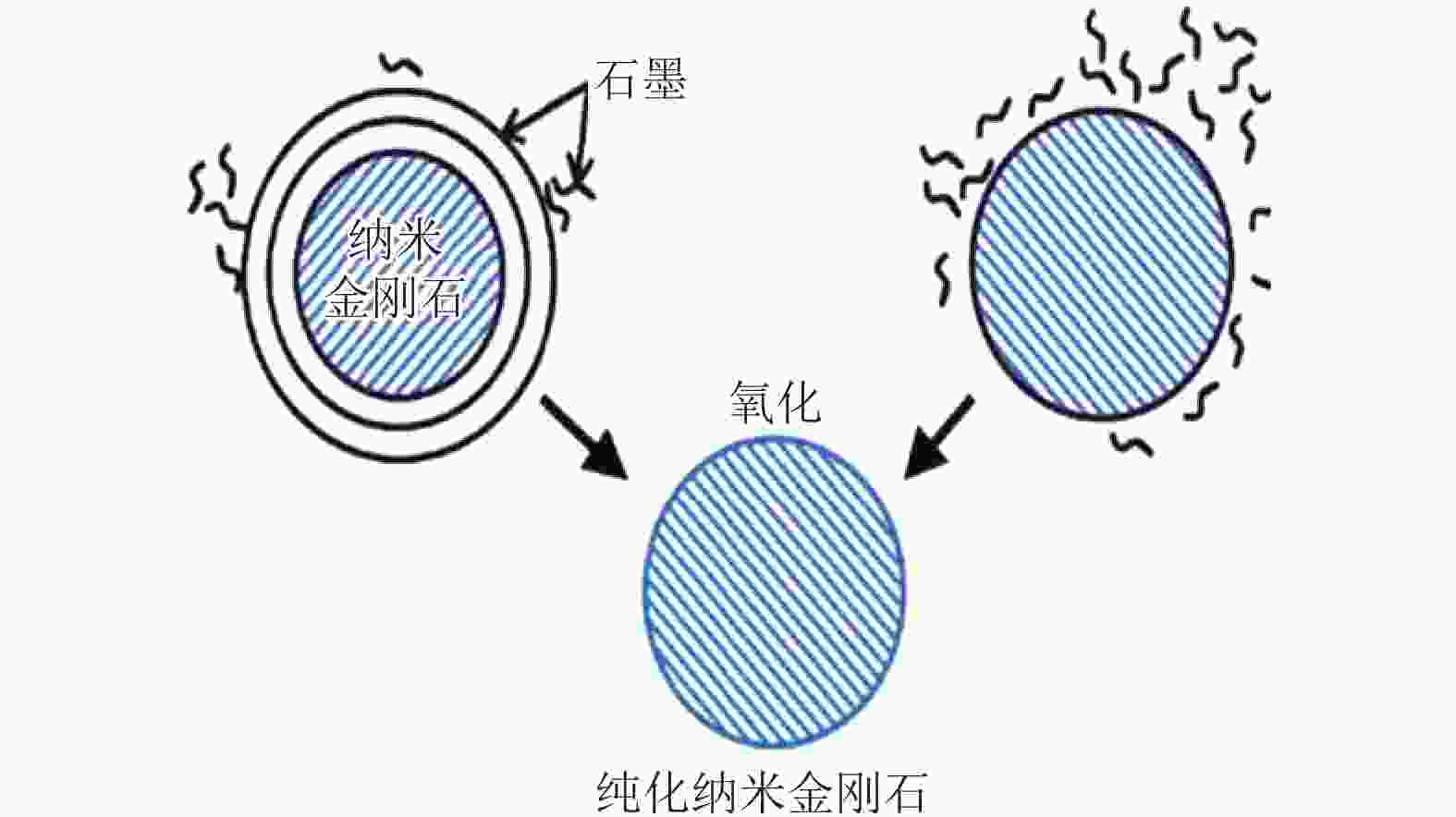
表 2 纳米金刚石氧化退火工艺
Table 2. Oxidation annealing process of the nanodiamonds
-
[1] Zou Q, Wang M Z, Wang Y H. Properties and application prospect of nanometer sized diamond. Diamond Abras Eng, 2003, 5(2): 54 doi: 10.3969/j.issn.1006-852X.2003.02.017邹芹, 王明智, 王艳辉. 纳米金刚石的性能与应用前景. 金刚石与磨料磨具工程, 2003, 5(2): 54 doi: 10.3969/j.issn.1006-852X.2003.02.017 [2] Daulton T L. Nanodiamonds in the cosmos: microstructural and trapped element isotopic data // Synthesis, Properties and Applications of Ultrananocrystalline Diamond. St. Petersburg, 2004: 49 [3] Wang F F, Peng J, Zhang L Q, et al. Preparation of nanodiamond by crushing method and research on its grading technology. Superhard Mater Eng, 2016, 28(5): 15 doi: 10.3969/j.issn.1673-1433.2016.05.004王芬芬, 彭进, 张琳琪, 等. 破碎法纳米金刚石的制备及其分级工艺研究. 超硬材料工程, 2016, 28(5): 15 doi: 10.3969/j.issn.1673-1433.2016.05.004 [4] Yang G W, Wang J B, Liu Q X. Preparation of nano-crystalline diamonds using pulsed laser induced reactive quenching. J Phys Condens Matter, 1998, 10(35): 7923 doi: 10.1088/0953-8984/10/35/024 [5] Frenklach M, Howard W, Huang D, et al. Induced nucleation of diamond powder. Appl Phys Lett, 1991, 59(5): 546 doi: 10.1063/1.105434 [6] Daulton T L, Kirk M A, Lewis R S, et al. Production of nanodiamonds by high-energy ion irradiation of graphite at room temperature. Nucl Instrum Methods Phys Res Sect B, 2001, 175(8): 12 [7] Banhart F, Ajayan P M. Carbon onions as nanoscopic pressure cells for diamond formation. Nature, 1996, 382(6590): 433 doi: 10.1038/382433a0 [8] Mochalin V N, Shenderova O, Ho D, et al. The properties and applications of nanodiamonds. Nat Nanotechnol, 2012, 7(1): 11 doi: 10.1038/nnano.2011.209 [9] Bondar V S, Puzyr A P. Use of nanodiamond particles for rapid isolation of recombinant apoobelin from escherichia coli. Dokl Biochem, 2000, 373(1-6): 129 [10] Vaijayanthimala V, Lee D K, Kim S V, et al. Nanodiamond-mediated drug delivery and imaging: challenges and opportunities. Expert Opin Drug Discovery, 2015, 12(5): 735 doi: 10.1517/17425247.2015.992412 [11] Nunn N, Torelli M, McGuire G, et al. Nanodiamond: a high impact nanomaterial. Curr Opin Solid State Mater Sci, 2017, 21(1): 1 doi: 10.1016/j.cossms.2016.06.008 [12] Kruger A, Kataoka F, Ozawa M, et al. Unusually tight aggregation in detonation nanodiamond: identification and disintegration. Carbon, 2005, 43(8): 1722 doi: 10.1016/j.carbon.2005.02.020 [13] Ozawa M, Inaguma M, Takahashi M, et al. Preparation and behavior of brownish, clear nanodiamond colloids. Adv Mater, 2007, 19(9): 1201 doi: 10.1002/adma.200601452 [14] Eidelman E D, Siklitsky V I, Sharonova L V, et al. A stable suspension of single ultrananocrystalline diamond particles. Diamond Relat Mater, 2005, 14(11): 1765 [15] Pentecost A, Gour S, Mochalin V, et al. Deaggregation of nanodiamond powders using salt- and sugar-assisted milling. ACS Appl Mater Interfaces, 2010, 2(11): 3289 doi: 10.1021/am100720n [16] Turcheniuk K, Trecazzi C, Deeleepojananan C, et al. Salt-assisted ultrasonic deaggregation of nanodiamond. ACS Appl Mater Interfaces, 2016, 8(38): 25461 doi: 10.1021/acsami.6b08311 [17] Khan M, Li T H, Zhao T K, et al. Comparative study of the ball milling and acid treatment of functionalized nanodiamond composites. Int J Refract Met Hard Mater, 2018, 73(1): 46 [18] Wang P, Zhu F, Wang Z Q. Effect of dispersion of nanodiamond in aqueous medium by ultrasonic and dispersants. Diamond Abras Eng, 2015, 35(2): 46王沛, 朱峰, 王志强. 超声波以及分散剂对纳米金刚石在水性介质中分散行为的影响. 金刚石与磨料磨具工程, 2015, 35(2): 46 [19] Cao R J, Lin C G, Sun L, et al. Agglomeration of superfine powder and dispersion methods. Powder Metall Technol, 2006, 24(6): 460 doi: 10.3321/j.issn:1001-3784.2006.06.015曹瑞军, 林晨光, 孙兰, 等. 超细粉末的团聚及其消除方法. 粉末冶金技术, 2006, 24(6): 460 doi: 10.3321/j.issn:1001-3784.2006.06.015 [20] Li Y, Li B X, Zhao M Y, et al. The dispersion behavior of nano-diamond in aqueous medium. Superhard Mater Eng, 2010, 22(2): 5 doi: 10.3969/j.issn.1673-1433.2010.02.002李颖, 李变晓, 赵盟月, 等. 水性介质中纳米金刚石分散行为研究. 超硬材料工程, 2010, 22(2): 5 doi: 10.3969/j.issn.1673-1433.2010.02.002 [21] Aleksenskiy A E, Eydelman E D, Vul A Y. Deagglomeration of detonation nanodiamonds. Nanosci Nanotechnol Lett, 2011, 3(1): 68 doi: 10.1166/nnl.2011.1122 [22] Guan B. Surface Functionalization and Applications of Nanodiamond and Boron-Doped Diamond Materials[Dissertation]. Beijing: Technical Institute of Physics and Chemistry CAS, 2009关波. 纳米金刚石及金刚石薄膜材料的功能化修饰及应用研究[学位论文]. 北京: 中国科学院理化技术研究所, 2009 [23] Pedroso-Santana S, Sarabia-Saínz A, Fleitas-Salazar N, et al. Deagglomeration and characterization of detonation nanodiamonds for biomedical applications. J Appl Biomed, 2017, 15(1): 15 doi: 10.1016/j.jab.2016.09.003 [24] Liu K K, Cheng C L, Chang C C, et al. Biocompatible and detectable carboxylated nanodiamond on human cell. Nanotechnology, 2007, 18(32): 5102 [25] Krüger A, Liang Y J, Jarre G, et al. Surface functionalisation of detonation diamond suitable for biological applications. J Mater Chem, 2006, 16(24): 2322 doi: 10.1039/B601325B [26] Miao W P, Ding Y L, Zhai L P, et al. Progress of research on nano-diamond dispersion. Diamond Abras Eng, 2019, 39(1): 18苗卫朋, 丁玉龙, 翟黎鹏, 等. 爆轰法合成纳米金刚石的分散技术研究进展. 金刚石与磨料磨具工程, 2019, 39(1): 18 [27] Williams O A, Hees J, Dieker C, et al. Size-dependent reactivity of diamond nanoparticles. ACS Nano, 2010, 4(8): 4824 doi: 10.1021/nn100748k [28] Tsubota T, Mihara S, Murakam N, et al. Chemical modification of diamond surface with linoleic acid by using benzoyl peroxide. Diamond Relat Mater, 2011, 20(4): 584 doi: 10.1016/j.diamond.2011.03.009 [29] Myllymäki V, Helsinki. Zeta Positive Hydrogenated Nanodiamond Power, Zetapositive Single Digit Hydrogenated Nanodiamond Dispersion, and Methods for Producing the Same: US Patent, US9884767 B2. 2018-02-06 [30] Krueger A, Lang D. Functionality is key: recent progress in the surface modification of nanodiamond. Adv Funct Mater, 2012, 22(5): 890 doi: 10.1002/adfm.201102670 [31] Osswald S, Yushin G, Mochalin V, et al. Control of sp2/sp3 carbon ratio and surface hemistry of nanodiamond powders by selective oxidation in air. J Am Chem Soc, 2006, 128(35): 11635 doi: 10.1021/ja063303n [32] Gaebel T, Bradac C, Chen J, et al. Size-reduction of nanodiamonds via air oxidation. Diamond Relat Mater, 2012, 21(10): 28 [33] Pichot V, Comet M, Fousson E, et al. An efficient purification method for detonation nanodiamonds. Diamond Relat Mater, 2008, 17(1): 13 doi: 10.1016/j.diamond.2007.09.011 [34] Li C C, Huang C L. Preparation of clear colloidal solutions of detonation nanodiamond in organic solvents. Colloids Surf A, 2010, 353(1): 52 doi: 10.1016/j.colsurfa.2009.10.019 [35] Xu K, Xue Q J. A new method for deaggregation of nanodiamond from explosive detonation: graphitization-oxidation method. Phys Solid State, 2004, 46(4): 649 doi: 10.1134/1.1711442 [36] Shenderova O, Petrov I, Walsh J, et al. Modification of detonation nanodiamonds by heat treatment in air. Diamond Relat Mater, 2006, 15(11): 1799 [37] Wang X H, Wang J, Li X J, et al. Study on stable dispersion of detonation nanodiamond in base oil. J Funct Mater, 2014, 45(20): 20060 doi: 10.3969/j.issn.1001-9731.2014.20.013王小红, 王俊, 李晓杰, 等. 爆轰纳米金刚石在基础油中稳定分散研究. 功能材料, 2014, 45(20): 20060 doi: 10.3969/j.issn.1001-9731.2014.20.013 [38] Mohan N, Tzeng Y K, Yang L, et al. Sub-20-nm fluorescent nanodiamonds as photostable biolabels and fluorescence resonance energy transfer donors. Adv Mater, 2010, 22(7): 843 doi: 10.1002/adma.200901596 [39] Chang Y R, Lee H Y, Chen K, et al. Mass production and dynamic imaging of fluorescent nanodiamonds. Nat Nanotechnol, 2008, 3(5): 284 doi: 10.1038/nnano.2008.99 [40] Gibson N, Shenderova O, Luo T J M, et al. Colloidal stability of modified nanodiamond particles. Diamond Relat Mater, 2009, 18(4): 620 doi: 10.1016/j.diamond.2008.10.049 [41] Kozak H, Artemenko A, Čermák J, et al. Oxidation and reduction of nanodiamond particles in colloidal solutions by laser irradiation or radio-frequency plasma treatment. Vib Spectrosc, 2016, 83(1): 108 [42] Girard H A, Arnault J C, Perruchas S, et al. Hydrogenation of nanodiamonds using MPCVD: a new route toward organic functionalization. Diamond Relat Mater, 2010, 19(7): 1117 [43] Ray M A, Shenderova O, Hook W, et al. Cold plasma functionalization of nanodiamond particles. Diamond Relat Mater, 2006, 15(11-12): 1809 doi: 10.1016/j.diamond.2006.06.003 [44] Zhang T P. Research of Deagglomeration of Detonation Nanodiamond by Laser Ablation and Its Mechanism of Fluorescence[Dissertation]. Tianjing: Tianjin University, 2009张泰平. 纳米金刚石的激光分散及发光机理研究[学位论文]. 天津: 天津大学, 2009 [45] Xu X Y, Zhu Y W, Wang B C. Study on the deagglomeration of nanodiamonds in water-based systems // Progress of Nanostructured Materials and Technical Applications: Proceedings of the 3rd National Conference on Nanomaterials and Technology Applications. Nanjing, 2003: 195许向阳, 朱永伟, 王柏春. 水基体系中纳米金刚石的解团聚研究//纳米材料和技术应用进展—全国第三届纳米材料和技术应用会议论文集. 南京, 2003: 195 [46] Liang Y, Meinhardt T, Jarre G, et al. Deagglomeration and surface modification of thermally annealed nanoscale diamond. J Colloid Interface Sci, 2011, 354(1): 23 doi: 10.1016/j.jcis.2010.10.044 [47] Krueger A, Stegk J, Liang Y J, et al. Biotinylated nanodiamond: simple and efficient functionalization of detonation diamond. Langmuir, 2008, 24(8): 4200 doi: 10.1021/la703482v [48] Kuznetsov O, Sun Y Q, Thaner R, et al. Water-soluble nanodiamond. Langmuir, 2012, 28(11): 5243 doi: 10.1021/la204660h [49] Beyler-Çiğil A, Kahraman M V. Effect of surface modification on nano-diamond particles for surface and thermal property of UV-curable hybrid coating. Prog Org Coat, 2016, 101(10): 468 [50] Zhang Q X, Naito K, Tanaka Y, et al. Grafting polyimides from nanodiamonds. Macromolecules, 2008, 41(3): 536 doi: 10.1021/ma702268x [51] Ma W J, Yu X Y, Qu X W, et al. Functionalization of agglomerating nanodiamonds with biodegradable poly(ε-caprolactone) through surface-initiated polymerization. Diamond Relat Mater, 2016, 62: 14 doi: 10.1016/j.diamond.2015.12.011 [52] Hu H X, Guo H M, Yu X Y, et al. Surface modification and disaggregation of detonation nanodiamond particles with biodegradable polyurethane. Colloids Surf A, 2019, 563(20): 302 -




 下载:
下载: