-
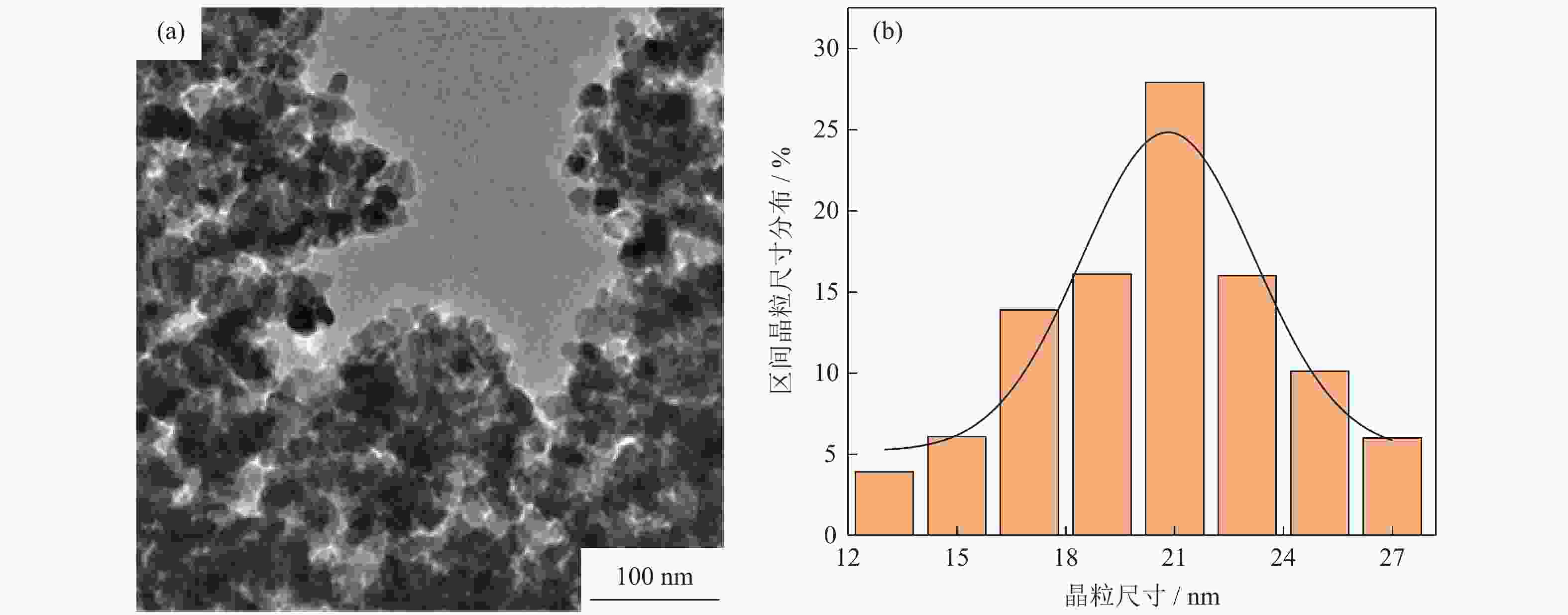
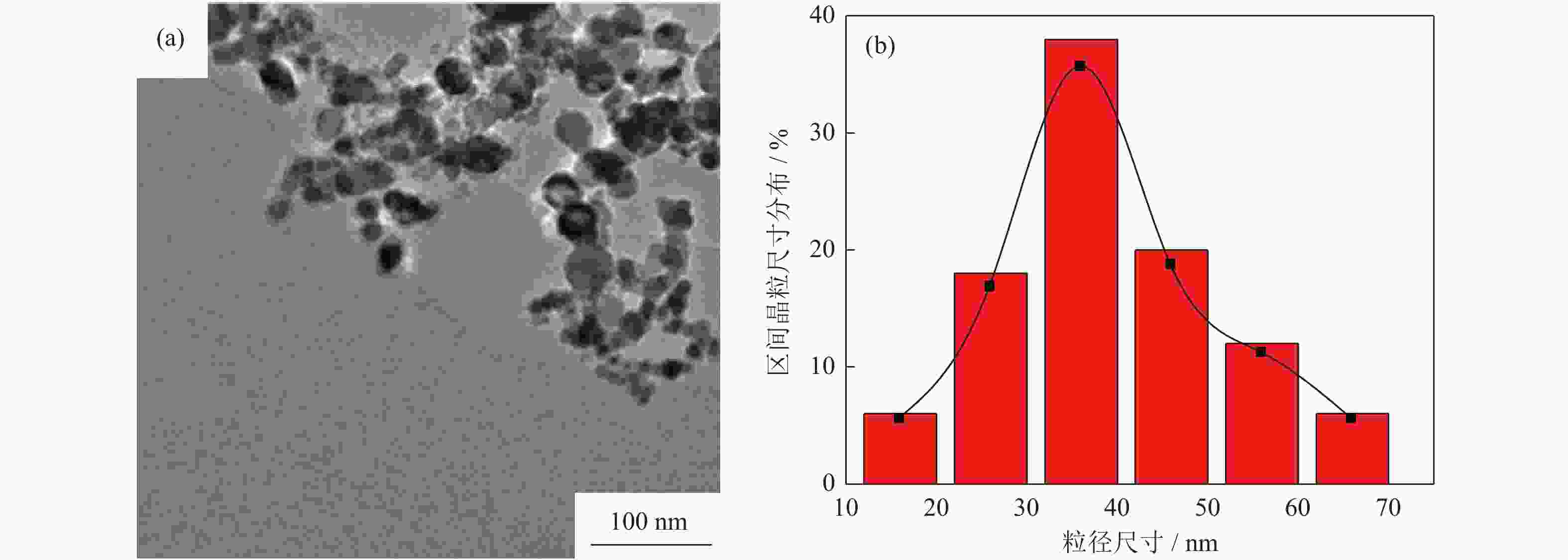
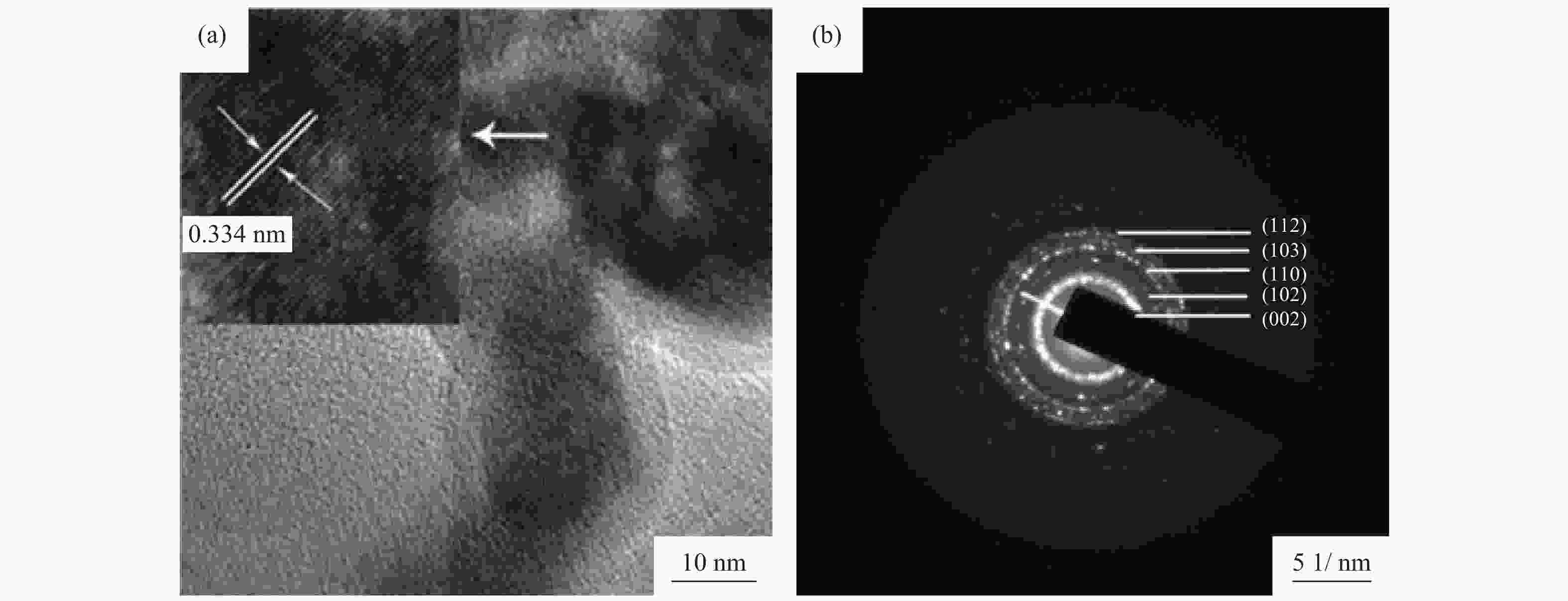
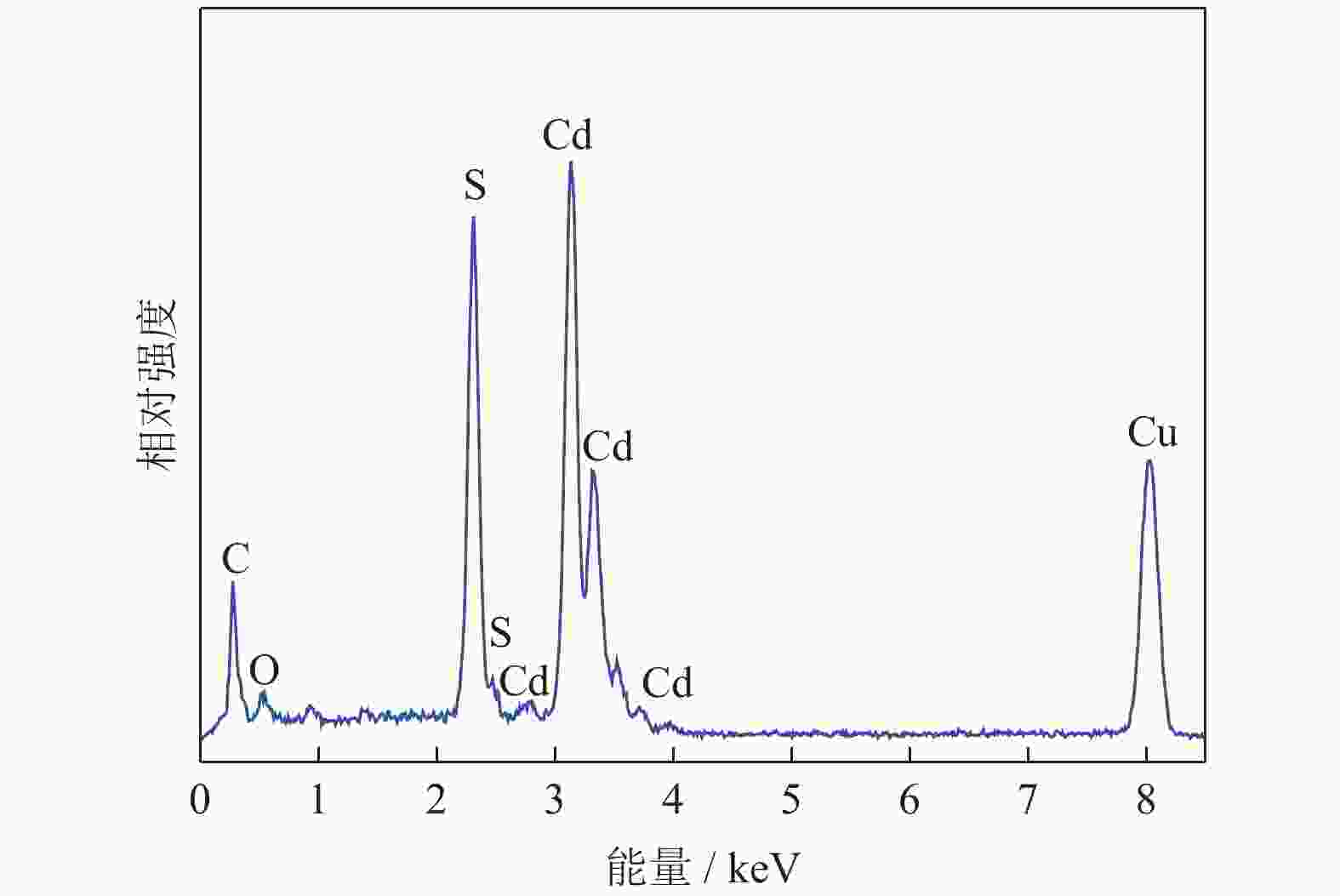
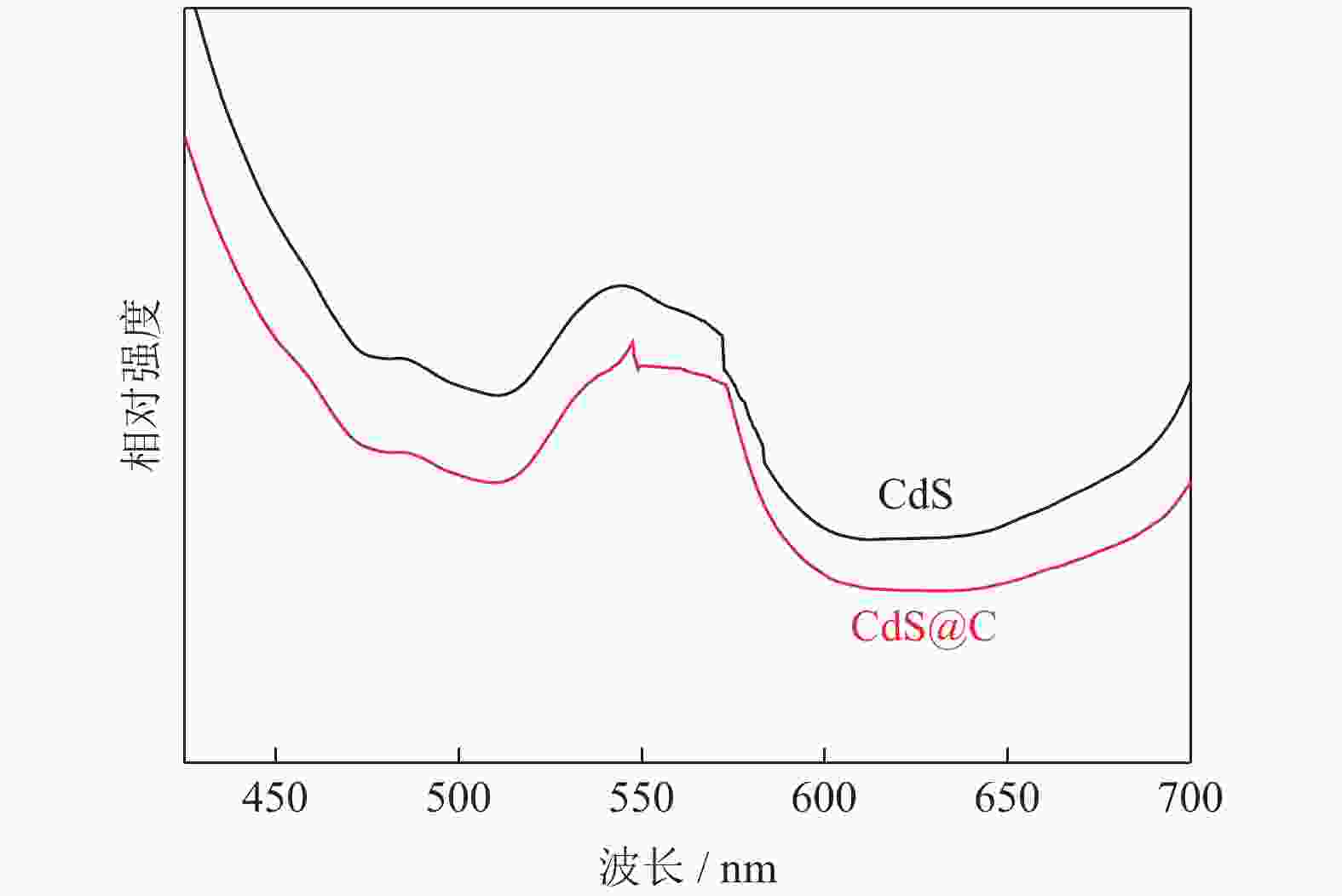
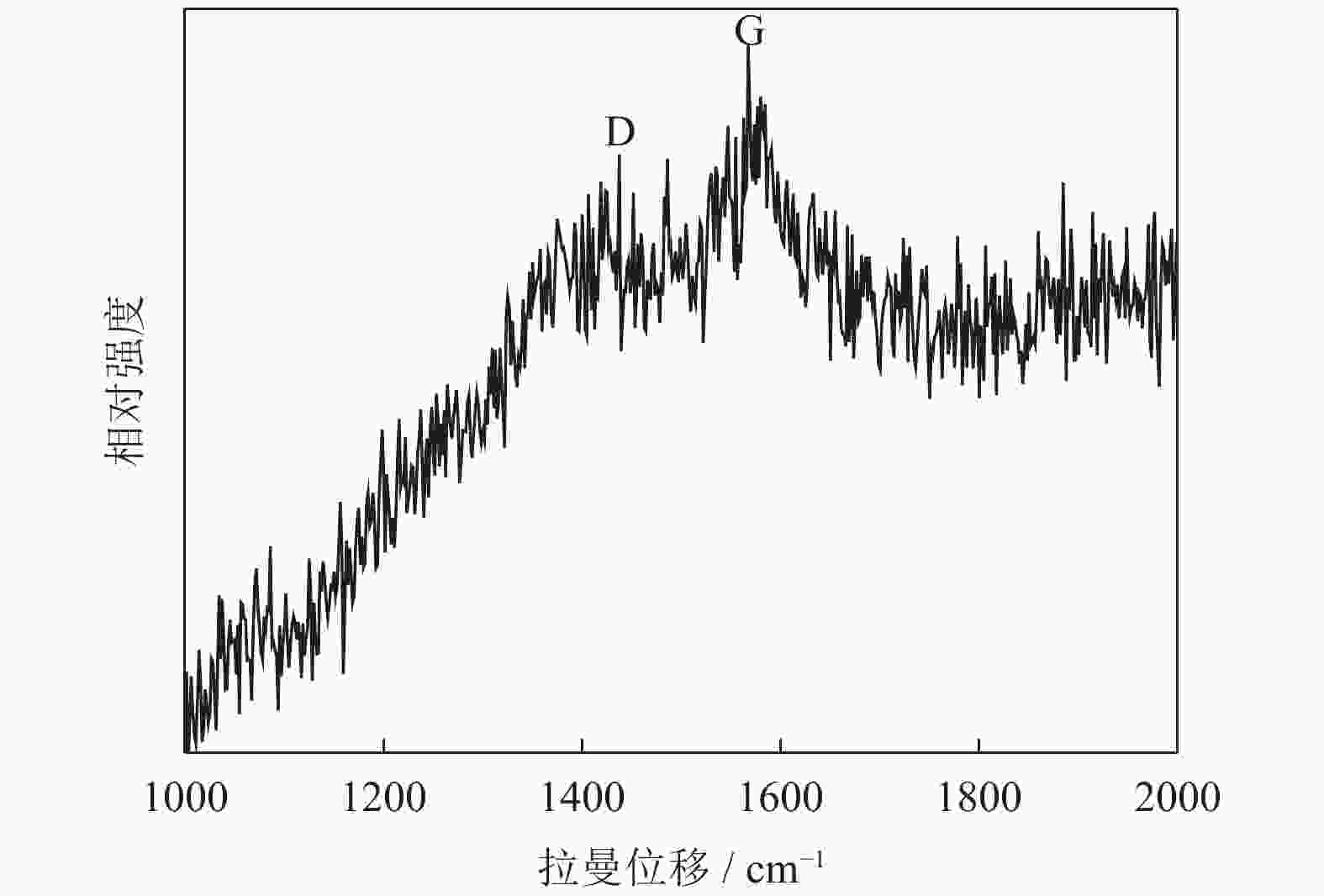
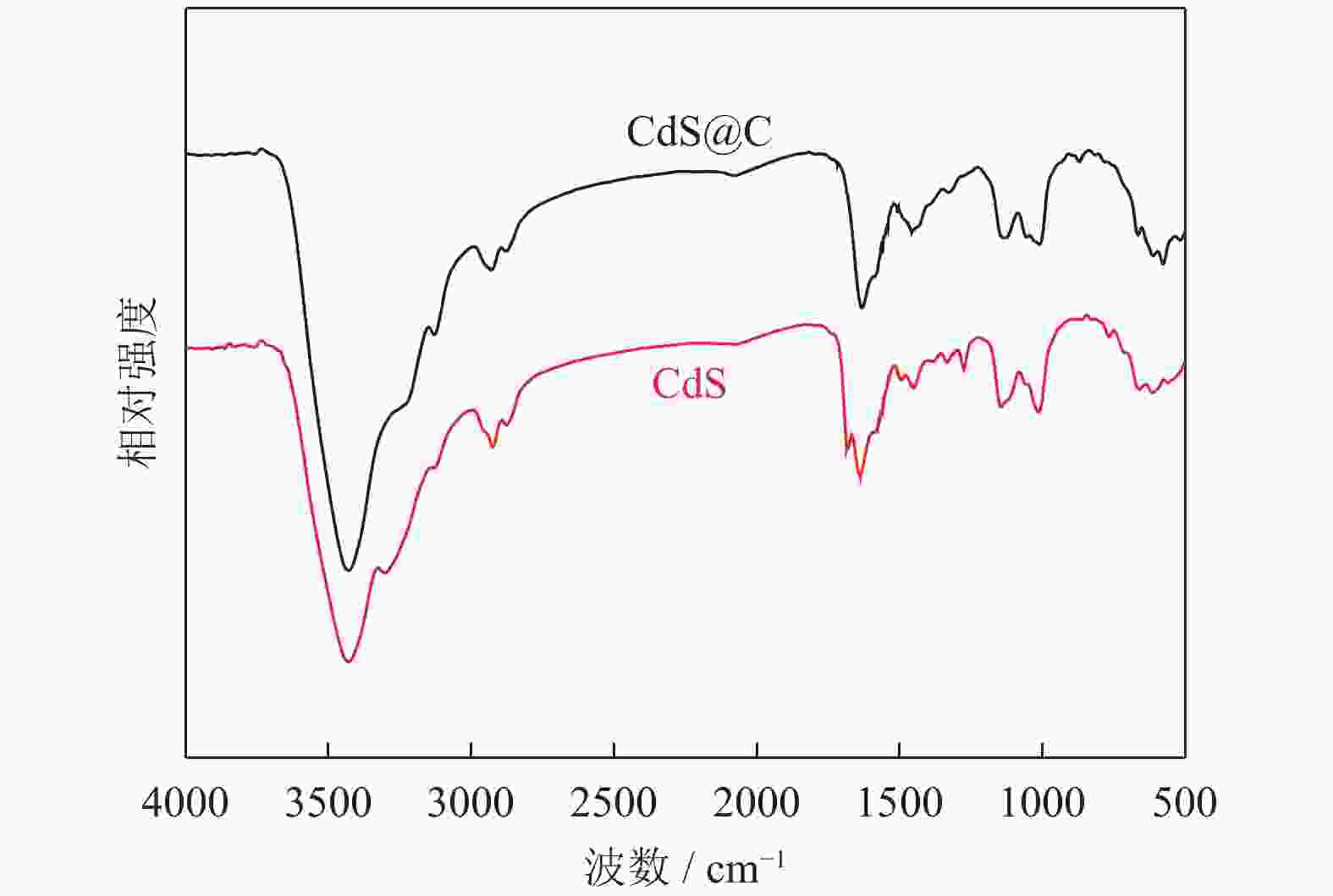
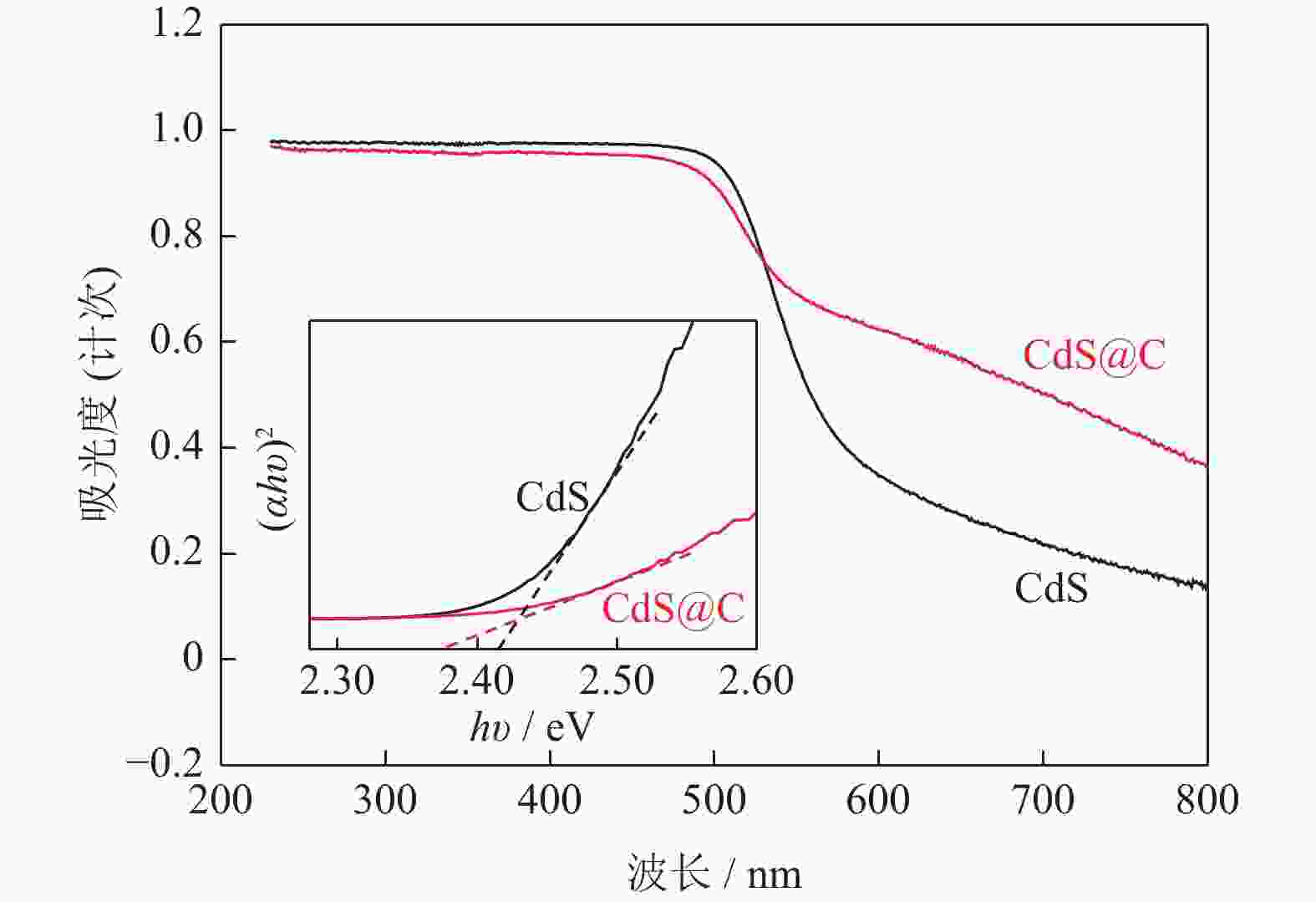
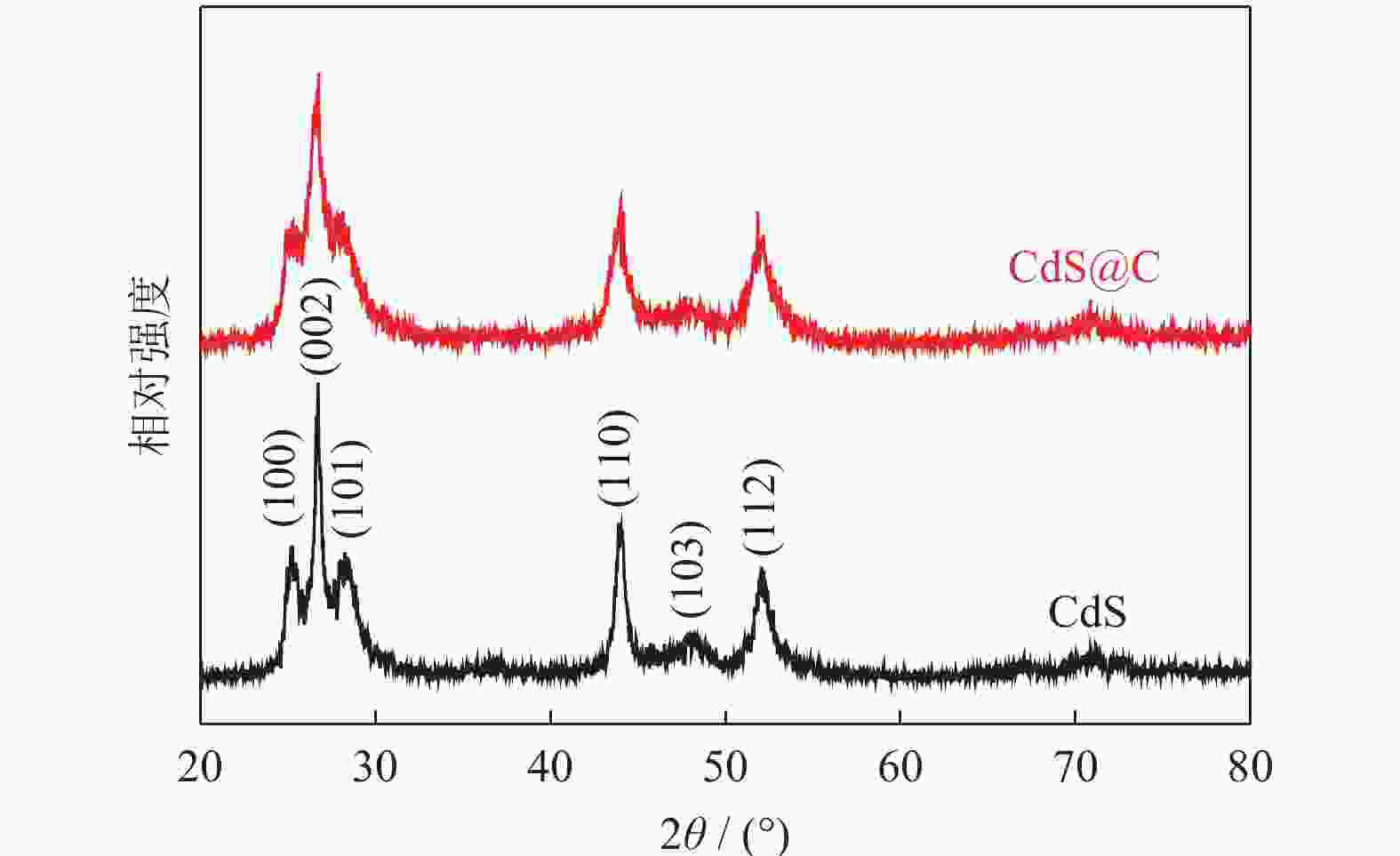
摘要: 采用水热法制备了核壳结构的碳包覆CdS纳米颗粒(CdS@C),利用X射线衍射、高分辨透射电子显微镜和光谱仪研究了碳包覆层对CdS@C的微观结构与光学性能影响。结果表明,碳包覆CdS纳米颗粒具有明显的核壳结构,内核为六方纤锌矿结构CdS,外壳为碳层。颗粒形貌主要为球形或椭球形结构,粒度均匀,分散性良好,粒径分布在20~60 nm范围,平均粒径为35 nm,外壳碳层的厚度为5 nm。光致发光光谱显示碳包覆CdS的发光强度比纯CdS弱,Raman光谱表明碳包覆CdS样品中碳包覆层的石墨化程度较低,傅里叶变换红外光谱表明纯CdS和碳包覆CdS样品的吸收峰位基本相近,紫外‒可见吸收光谱表明碳包覆CdS纳米晶提高了可见光吸收强度,能隙变窄,发生了红移现象。Abstract: Carbon encapsulated CdS nanoparticles with the core-shell structure were successfully prepared by hydrothermal method, and the influences of carbon coating layer on the microstructure and optical properties of CdS@C were investigated by X-ray diffraction, high-resolution transmission electron microscope, and spectroscope. The results show that, the carbon encapsulated CdS nanoparticles have the obvious core-shell structure, the inner core is CdS with the hexagonal wurtzite structure, and the outer shell is carbon layer. The particle morphology is mainly spherical or ellipsoidal shape with the uniform particle size and good dispersion, the particle size distribution is in the range of 20~60 nm, the average particle size is 35 nm, and the thickness of carbon layer is about 5 nm. Photoluminescence spectra show that the luminescence intensity of the carbon encapsulated CdS is weaker than that of the pure CdS, the Raman spectra reveal that the carbon coating layer shows the low graphitization degree in the carbon encapsulated CdS samples, the Fourier transform infrared spectra show that the absorption peaks of the pure CdS and the carbon encapsulated CdS samples are basically similar. The ultraviolet visible spectra indicate that the carbon encapsulated CdS nanocrystals enhance the absorption intensity of the visible light, narrow the energy gap, and result in the red shift.
-
Key words:
- carbon-encapsulated /
- CdS /
- core-shell structure /
- microstructure /
- optical properties
-
表 1 CdS和CdS@C样品的X射线衍射测算结果
Table 1. XRD results of CdS and CdS@C samples
样品 2θ / (°) 半峰宽 晶面间距 / nm 晶粒尺寸 / nm 晶格常数 / nm CdS 26.642 0.398 0.334 20.28 a=41.096,c=66.812 CdS@C 26.576 0.487 0.335 16.57 a=41.007,c=67.547 -
[1] Yang X D, Wang Z S, Lü X Z, et al. Enhanced photocatalytic activity of Zn-doped dendritic-like CdS structures synthesized by hydrothermal synthesis. J Photochem Photobiol A, 2016, 329: 175 doi: 10.1016/j.jphotochem.2016.07.005 [2] Liu Y D, Ren L, Qi X, et al. One-step hydrothermal fabrication and enhancement of the photocatalytic performance of CdMoO4/CdS hybrid materials. RSC Adv, 2014, 4(17): 8772 doi: 10.1039/c3ra46051g [3] Liu I P, Chen L Y, Lee Y L. Effect of sodium acetate additive in successive ionic layer adsorption and reaction on the performance of CdS quantum-dot-sensitized solar cells. J Power Sources, 2016, 325: 706 doi: 10.1016/j.jpowsour.2016.06.095 [4] Fan K, Liao C, Xu R L, et al. Effect of shell thickness on electrochemical property of wurtzite CdSe/CdS core/shell nanocrystals. Chem Phys Lett, 2015, 633: 1 doi: 10.1016/j.cplett.2015.05.006 [5] Yang H, Kershaw S V, Wang Y, et al. Shuttling photoelectrochemical electron transport in tricomponent CdS/rGO/TiO2 nanocomposites. J Phys Chem C, 2013, 117(40): 20406 doi: 10.1021/jp405227t [6] Ma B, Xu H, Lin K, et al. Mo2C as non-noble metal Co-catalyst in Mo2C/CdS composite for enhanced photocatalytic H2 evolution under visible light irradiation. Chemsuschem, 2016, 9(8): 820 doi: 10.1002/cssc.201501652 [7] Yan X, Wu Z, Huang C, et al. Hydrothermal synthesis of CdS/CoWO4, heterojunctions with enhanced visible light properties toward organic pollutants degradation. Ceram Int, 2017, 43(7): 5388 doi: 10.1016/j.ceramint.2016.12.060 [8] Kumar S, Mehta S K. Varying photoluminescence emission of CdS nanoparticles in aqueous medium: A comparative study on effect of surfactant structure. Nano-Structures Nano-Objects, 2015, 2: 1 doi: 10.1016/j.nanoso.2015.05.002 [9] Zirak M, Akhavan O, Moradlou O, et al. Vertically aligned ZnO@CdS nanorod heterostructures for visible light photoinactivation of bacteria. J Alloys Compd, 2014, 590(4): 507 [10] Yang H, Jin Z, Fan K, et al. The roles of Ni nanoparticles over CdS nanorods for improved photocatalytic stability and activity. Superlattices Microstruct, 2017, 111: 687 doi: 10.1016/j.spmi.2017.07.025 [11] Wang Y F, Chen W, Chen X, et al. Effect of sulfur source on photocatalytic degradation performance of CdS/MoS2 prepared with one-step hydrothermal synthesis. J Environ Sci, 2018, 65(3): 347 [12] Jo W K, Selvam N C S. Fabrication of photostable ternary CdS/MoS2/MWCNs hybrid photocatalysts with enhanced H2, generation activity. Appl Catal A, 2016, 525: 9 doi: 10.1016/j.apcata.2016.06.036 [13] Zhang Z, Ren Y, Han L, et al. Mixed-solvothermal synthesis of CdS micro/nanostructures with optical and ferromagnetic properties. Physica E, 2017, 92: 30 doi: 10.1016/j.physe.2017.04.027 [14] Zou S, Fu Z, Xiang C, et al. Mild, one-step hydrothermal synthesis of carbon-coated CdS nanoparticles with improved photocatalytic activity and stability. Chin J Catal, 2015, 36(7): 1077 doi: 10.1016/S1872-2067(15)60827-0 [15] Li D, Xie J, Zhang Y, et al. Convenient synthesis of magnetically recyclable Fe3O4@C@CdS photocatalysts by depositing CdS nanocrystals on carbonized ferrocene. J Alloys Compd, 2015, 646: 978 doi: 10.1016/j.jallcom.2015.06.075 [16] Hu Y, Gao X, Yu L, et al. Carbon-coated CdS petalous nanostructures with enhanced photostability and photocatalytic activity. Angew Chem Int Ed, 2013, 52(21): 5636 doi: 10.1002/anie.201301709 [17] Liu Y, Yu Y X, Zhang W D. Carbon quantum dots-doped CdS microspheres with enhanced photocatalytic performance. J Alloys Compd, 2013, 569(9): 102 [18] Patel J D, Vu T T D, Mighri F. Preparation and characterization of CdS coated multiwalled carbon nanotubes. Mater Lett, 2017, 196: 161 doi: 10.1016/j.matlet.2017.03.046 [19] Wang F, Liang L, Chen K, et al. CO2, induced template approach to fabricate the porous C/CdS visible photocatalyst with superior activity and stability. J Mol Catal A Chem, 2016, 425: 76 doi: 10.1016/j.molcata.2016.09.034 [20] Chen R, Han B, Yang L, et al. Controllable synthesis and characterization of CdS quantum dots by a microemulsion-mediated hydrothermal method. J Lumin, 2016, 172: 197 doi: 10.1016/j.jlumin.2015.12.006 [21] Zhang J M, Wang J, Zhang J, et al. Preparation of graphene oxide composites and study on adsorption properties of copper ions. Powder Metall Technol, 2018, 36(6): 445张建民, 王晶, 张继, 等. 氧化石墨烯复合材料的制备及对铜离子吸附性能的研究. 粉末冶金技术, 2018, 36(6): 445 [22] Li L, Dong G X, Li Z F, et al. Effects of solid phase reaction conditions on electrochemical performance of lithium iron phosphate. Powder Metall Technol, 2019, 37(5): 332李雷, 董桂霞, 李宗峰, 等. 固相反应条件对磷酸铁锂电化学性能的影响. 粉末冶金技术, 2019, 37(5): 332 [23] Wei B Z, Chen W C, Zhu X, et al. Study of electroless plating Cu by reduced graphene oxide and the effects on the microstructures and properties of RGO/Cu composites. Powder Metall Technol, 2018, 36(5): 363魏邦争, 陈闻超, 朱曦, 等. 石墨烯化学镀铜及其对石墨烯/铜基复合材料组织性能的影响. 粉末冶金技术, 2018, 36(5): 363 [24] Zou S, Fu Z H, Zeng M, et al. Study on preparation of CuS/CdS photocatalysts and their photocatalytic performance. J Nat Sci Hunan Normal Univ, 2016, 39(5): 57 doi: 10.7612/j.issn.1000-2537.2016.05.009邹帅, 伏再辉, 曾明, 等. CuS/CdS光催化剂的制备及其光催化性能研究. 湖南师范大学自然科学学报, 2016, 39(5): 57 doi: 10.7612/j.issn.1000-2537.2016.05.009 [25] Zhou M, Hu Y, Liu Y, et al. Microwave-assisted route to fabricate coaxial ZnO/C/CdS nanocables with enhanced visible light-driven photocatalytic activity. Crystengcomm, 2012, 14(22): 7686 doi: 10.1039/c2ce25540e -



 下载:
下载: