Preparation of silver-coated transition metal selenides and properties of silver-based composites
-
摘要: 为了提升银基复合材料中各相与基体之间的界面结合强度,改善材料性能,采用化学还原制备银包覆NbSe2及银包覆Ti0.09Nb0.91Se2颗粒,并通过粉末冶金法制备了Ag基复合材料。结果表明:银颗粒均匀分布在过渡族金属硒化物表面,包覆效果好。银包覆处理增加了过渡族金属硒化物与金属基体Ag之间的接触,改善了Ag与过渡族金属硒化物之间的润湿性,减小了Ag与过渡族金属颗粒之间的排斥力,使得过渡族金属硒化物在Ag基复合材料发生分解的可能性大大降低,提高了复合材料的力学性能,增强了过渡族金属硒化物与基体之间的界面结合强度。与含有NbSe2的银基复合材料相比,含有Ag包覆过渡族金属硒化物的银基复合材料的摩擦性能略有下降。Abstract: To improve the interface bonding strength between the substrate and phases in the Ag-based composite, the silver-coated NbSe2 and silver-coated Ti0.09Nb0.91Se2 particles were synthesized by chemical reduction method, and the Ag-based composites were prepared by powder metallurgy. The results show that, the silver particles are uniformly distributed on the surface of the transition metal selenides, and the Ti0.09Nb0.91Se2 particles show the better coating effect. After covering the Ag coating, the contact between the transition metal selenides and the metal matrix silver is increased, leading to the better wettability and the smaller repulsive force between Ag and the transition metal selenide. Moreover, the possibility of the transition metal selenide decomposition in the Ag-based composite materials is greatly reduced after the Ag coating. The densification of the Ag-based composite materials is improved and the interface bonding strength between the Ag substrates and the transition metal selenides is enhanced. Compared with the Ag-based composites with the uncoated NbSe2, the friction properties of the Ag-based composites containing the silver-coated transition metal selenides decrease slightly.
-
Key words:
- transition metal selenides /
- composites /
- interface strength /
- tribological properties
-
图 2 Ag/NbSe2和Ag/Ti0.09Nb0.91Se2扫描电子显微形貌和能谱分析:(a)Ag/NbSe2显微形貌;(b)Ag/NbSe2能谱分析;(c)Ag/Ti0.09Nb0.91Se2显微形貌;(d)Ag/Ti0.09Nb0.91Se2能谱分析
Figure 2. SEM images and EDS analysis of the Ag/NbSe2 and Ag/Ti0.09Nb0.91Se2: (a) SEM image of Ag/NbSe2; (b) EDS analysis of Ag/NbSe2; (c) SEM image of Ag/Ti0.09Nb0.91Se2; (d) EDS analysis of Ag/Ti0.09Nb0.91Se2
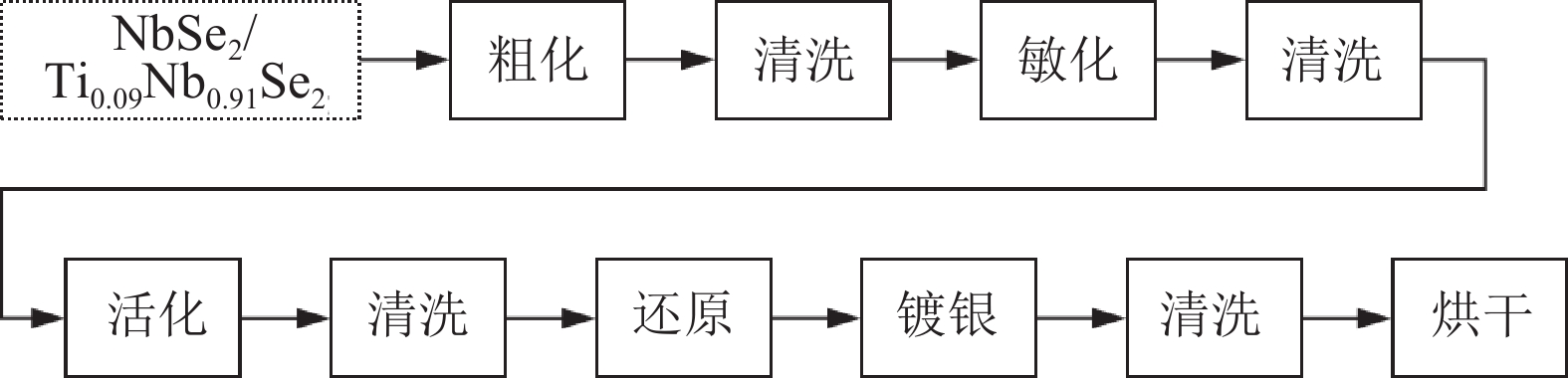
表 1 化学镀工艺中各种溶液的组成
Table 1. Composition of the various solution in the electroless plating process
溶液名称 药品名称 用量 配置方法 还原溶液 C2H5OH 150 mL 在C2H5OH中滴入HCHO,同时加入NaOH,
持续搅拌直到溶液变清澈即可。HCHO 10 mL NaOH 10 g 敏化溶液 SnCl2 12 g 将SnCl2加入HCL中,保持搅拌至完全溶解,再加入到
去离子水中,保持1 h,即可得到敏化溶液。HCL 16 mL 去离子水 200 mL 银氨溶液 AgNO3 9 g 将AgNO3溶于去离子水中,搅拌过程中缓慢加入NH3·H2O,
直至NH3·H2O全部加入即可。NH3·H2O 75 mL 去离子水 200 mL 表 2 Ag基复合材料试样化学成分(质量分数)
Table 2. Chemical compositions of the silver-based composites
% 试样 银粉 铬粉 Ag/Ti0.09Nb0.91Se2 Ag/NbSe2 纯NbSe2 Sa1 75 10 15 0 0 Sa2 75 10 0 15 0 Sa3 75 10 0 0 15 表 3 Ag基复合材料密度和力学性能
Table 3. Density and mechanical properties of the silver-based composites
试样 密度 / (g·cm−3) 孔隙度 / % 微硬度,HV 断裂强度 / MPa Sa1 8.12 7.87 211 162 Sa2 8.01 8.07 204 159 Sa3 7.89 8.19 197 146 -
[1] Shi Q, Tang H, Zhu H, et al. Synthesis and tribological properties of Ti-doped NbSe2 nanoparticles. Chalcogenide Lett, 2014, 11(5): 199 [2] Yuan M, Zhu H J, Peng H H, et al. Synthesis and tribological properties of ferrous based composites containing TiSe2 particles. Powder Metall Technol, 2016, 34(2): 106袁梦, 朱和军, 彭红红, 等. 含纳米TiSe2的铁基复合材料的制备及摩擦学性能研究. 粉末冶金技术, 2016, 34(2): 106 [3] Shi Q, Yang J, Peng W X, et al. Synergetic effect of NbSe2 and Cr2Nb on the tribological and electrical behavior of Cu-based electrical contact composites. RSC Adv, 2015, 5(122): 100472 doi: 10.1039/C5RA17786C [4] Chen S. Preparation of NbS 2xSe 2(1‒x) and Tribology Property of Copper Based Composite Mixed NbS 2xSe 2(1‒x) [Dissertation]. Zhenjiang: Jiangsu University, 2014陈帅. NbS2 xSe2(1‒ x)材料制备及其Cu基复合材料摩擦学性能研究[学位论文]. 镇江: 江苏大学, 2014 [5] Shi Q. Research on Electrical Contact Composites Containing Transition Metal Selenides [Dissertation]. Zhenjiang: Jiangsu University, 2017施琴. 过渡族金属硒化物电接触复合材料的研究[学位论文]. 镇江: 江苏大学, 2017 [6] Anand T J S, Shariza S. A study on molybdenum sulphoselenide (MoS x Se2− x , 0≤ x≤2) thin films: Growth from solution and its properties. Electrochim Acta, 2012, 81: 64 doi: 10.1016/j.electacta.2012.07.077 [7] Rossnagel K. On the origin of charge-density waves in select layered transition-metal dichalcogenides. J Phys Condens Matter, 2011, 23(21): 213001 doi: 10.1088/0953-8984/23/21/213001 [8] Bhatt R, Basu R, Bhattacharya S, et al. Low temperature thermoelectric properties of Cu intercalated TiSe2: a charge density wave material. Appl Phys A, 2013, 111(2): 465 doi: 10.1007/s00339-012-7536-8 [9] Moustafa S F, El-Badry S A, Sanad A M, et al. Friction and wear of copper–graphite composites made with Cu-coated and uncoated graphite powders. Wear, 2002, 253(7-8): 699 doi: 10.1016/S0043-1648(02)00038-8 [10] Cui G, Bi Q, Zhu S, et al. Tribological properties of bronze-graphite composites under sea water condition. Tribol Int, 2012, 53(9): 76 [11] Wei B Z, Chen W C, Zhu X, et al. Study of electroless plating Cu by reduced graphene oxide and the effects on the microstructures and properties of RGO/Cu composites. Powder Metall Technol, 2018, 36(5): 363 doi: 10.19591/j.cnki.cn11-1974/tf.2018.05.008魏邦争, 陈闻超, 朱曦, 等. 石墨烯化学镀铜及其对石墨烯/铜基复合材料组织性能的影响. 粉末冶金技术, 2018, 36(5): 363 doi: 10.19591/j.cnki.cn11-1974/tf.2018.05.008 [12] Xiong X, Chen J, Yao P P, et al. Effect of MoS2 on the sintering behaviors and mechanical properties of iron-based friction materials. Powder Metall Technol, 2006, 24(3): 182熊翔, 陈洁, 姚屏萍, 等. MoS2对铁基摩擦材料烧结行为及力学性能的影响. 粉末冶金技术, 2006, 24(3): 182 [13] Song J, Xu L, Xing R, et al. Ag nanoparticles coated NiO nanowires hierarchical nanocomposites electrode for nonenzymatic glucose biosensing. Sens Actuators B, 2013, 182: 675 doi: 10.1016/j.snb.2013.03.069 [14] Chee S S, Lee J H. Preparation and oxidation behavior of Ag-coated Cu nanoparticles less than 20 nm in size. J Mater Chem C, 2014, 2(27): 5372 doi: 10.1039/C4TC00509K [15] Li Y, Liu R, Zhang J, et al. Fabrication and microstructure of W‒Cu composites prepared from Ag-coated Cu powders by electroless plating. Surf Coat Technol, 2019, 361: 302 doi: 10.1016/j.surfcoat.2019.01.030 -




 下载:
下载: